IADR Abstract Archives
Are Fast-Setting Calcium Silicate Cements Biocompatiblewith Vital Pulp Therapy?
Objectives: MTA has been considered the gold standard in biomaterials for vital pulp therapy. However, it requires long duration of setting after application. To overcome the drawbacks of MTA, several calcium silicate cements are developed. The purpose of this study was assess the biocompatibility and bioactivity of newly developed calcium silicate cements.
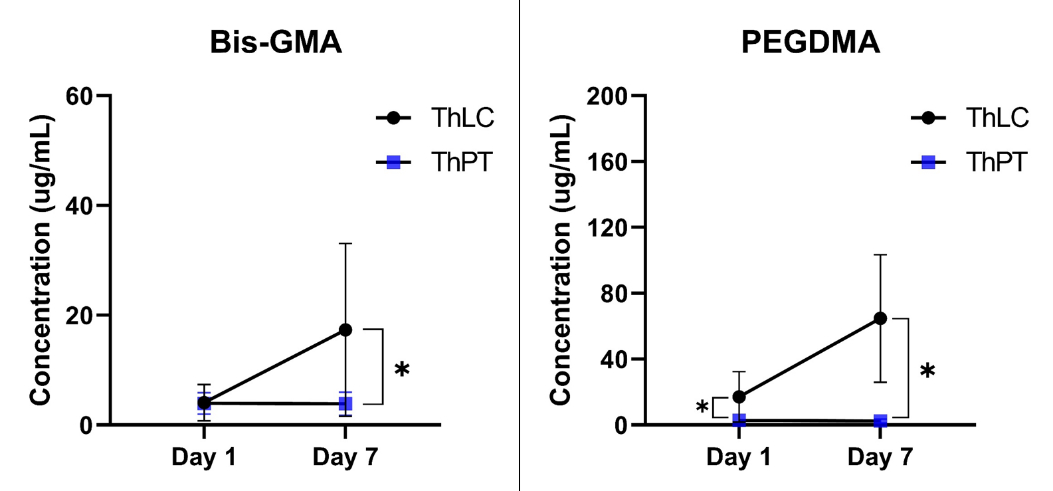
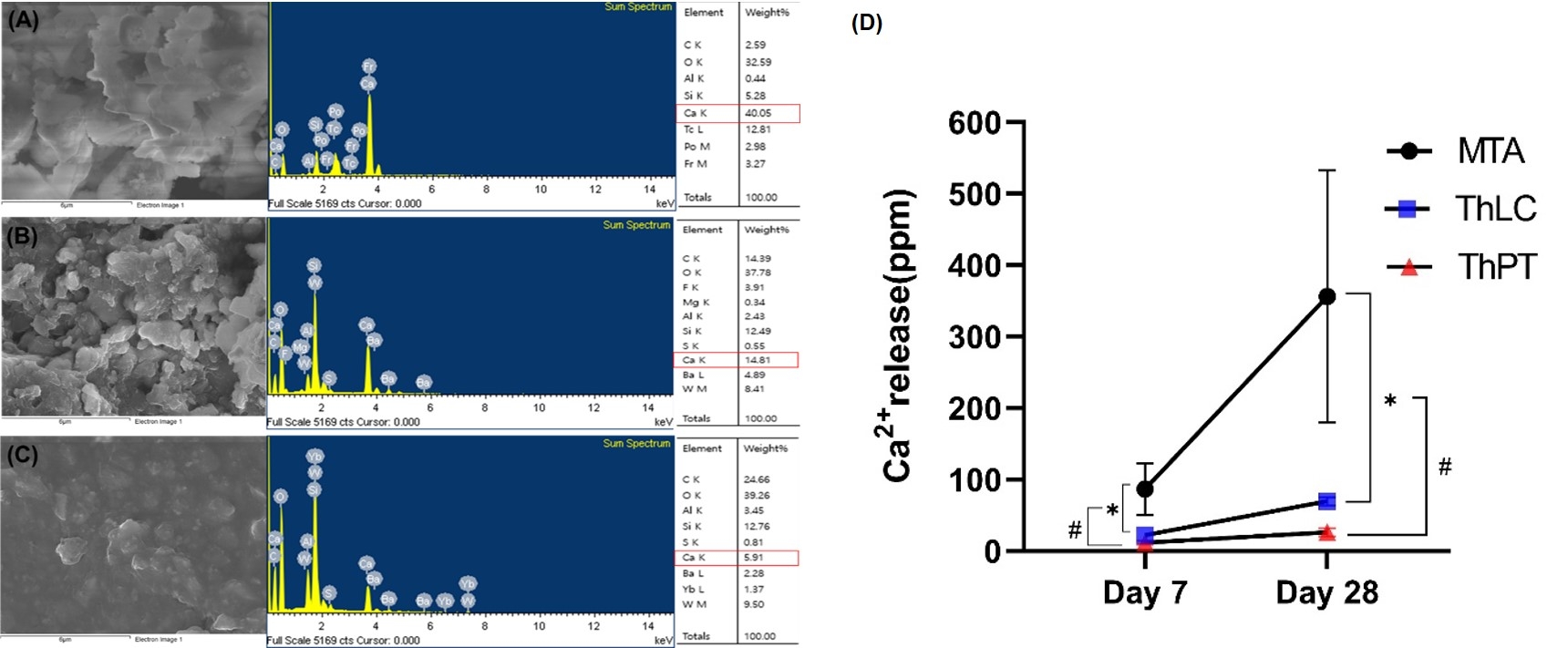
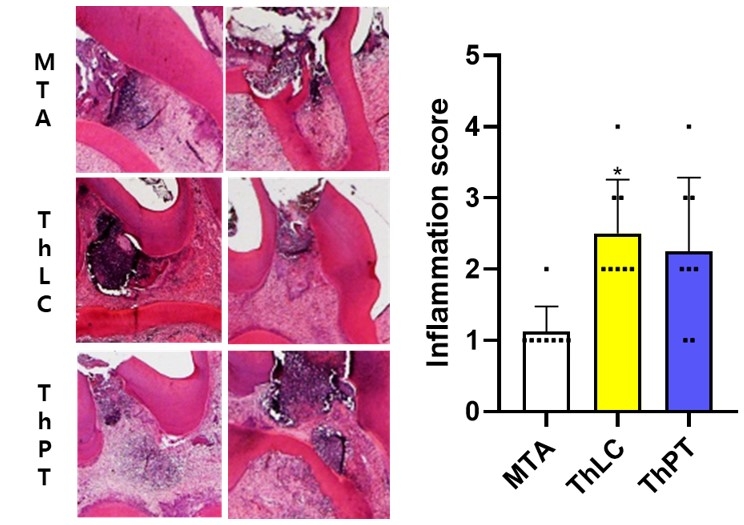
Methods: In-vitro samples of ProRoot MTA white (control), TheraCal LC, and TheraCal PT were prepared. Release of residual monomer (Bis-GMA and PEGDMA) was assessed using LC-MS/MS. Also, SEM/EDS mapping and ICP-OES analysis was performed to evaluate Ca2+ weight volume and release. Next, a direct pulp capping model was employed to the maxillary molars from Sprague-Dawley rats (n =8 per group). Intentional pulp exposure was made on the occlusal surface of the tooth. Then the exposure site was disinfected by 2% chlorohexidine. After confirmation of hemostasis, the materials were applied and the cavity was sealed. The animals were sacrificed either 7 or 28 days after the experiment. Histological analysis was performed to evaluate in-vivo biocompatibility and bioactivity.
Results: LC-MS/MS assay demonstrated that release of residual monomers was significantly lower in TheraCal PT compared to TheraCal LC (p < 0.05). Ca2+ release was significantly higher in TheraCal LC compared to TheraCal PT (p < 0.05). In-vivo histological analysis showed that there were no significant differences in inflammation score between TheraCal LC and TheraCal PT. However, TheraCal LC showed a significantly higher inflammation score compared to MTA (p < 0.017).
Conclusions: Within the limits of this study, TheraCal PT and TheraCal LC were biocompatible and bioactive for vital pulp therapy.
Methods: In-vitro samples of ProRoot MTA white (control), TheraCal LC, and TheraCal PT were prepared. Release of residual monomer (Bis-GMA and PEGDMA) was assessed using LC-MS/MS. Also, SEM/EDS mapping and ICP-OES analysis was performed to evaluate Ca2+ weight volume and release. Next, a direct pulp capping model was employed to the maxillary molars from Sprague-Dawley rats (n =8 per group). Intentional pulp exposure was made on the occlusal surface of the tooth. Then the exposure site was disinfected by 2% chlorohexidine. After confirmation of hemostasis, the materials were applied and the cavity was sealed. The animals were sacrificed either 7 or 28 days after the experiment. Histological analysis was performed to evaluate in-vivo biocompatibility and bioactivity.
Results: LC-MS/MS assay demonstrated that release of residual monomers was significantly lower in TheraCal PT compared to TheraCal LC (p < 0.05). Ca2+ release was significantly higher in TheraCal LC compared to TheraCal PT (p < 0.05). In-vivo histological analysis showed that there were no significant differences in inflammation score between TheraCal LC and TheraCal PT. However, TheraCal LC showed a significantly higher inflammation score compared to MTA (p < 0.017).
Conclusions: Within the limits of this study, TheraCal PT and TheraCal LC were biocompatible and bioactive for vital pulp therapy.