IADR Abstract Archives
Use of KDM4B Inhibitors to Target Periodontal Disease Progression
Objectives: The primary objective of this research is to develop small molecule inhibitors of KDM4B as adjunctive therapeutics for PD.
Methods: KDM4B protein was quantified in both healthy and diseased human oral epithelium using immunohistochemistry. Twelve-week old C57BL/6 mice were evaluated in a 5-day calvarial PD model using live Aggregatibacter actinomycetemcomitans (A.a) injections. Tissue sections were stained using specific antibodies for TRAP, F4/80 and KDM4B. Bone-marrow-derived-macrophages (BMDMs) were isolated from 12-week old C57BL/6 male and female mice and pre-treated with the KDM4B inhibitor, ML324. Pre-treated BMDMs were challenged with Aggregatibacter actinomycetemcomitans lipopolysaccharide (A.a LPS) for 8 and 24 hours, after which RNA and supernatant proteins were collected for q-RT-PCR and ELISA analysis. RAW264.7 cells were subject to the same treatment and stained for methylation markers using the appropriate fluorescent antibodies. Computational docking experiments using the ZINC database were screened to identify potential KDM4B inhibitors for optimization. Statistical significance was determined using unpaired t-tests (p<0.05).
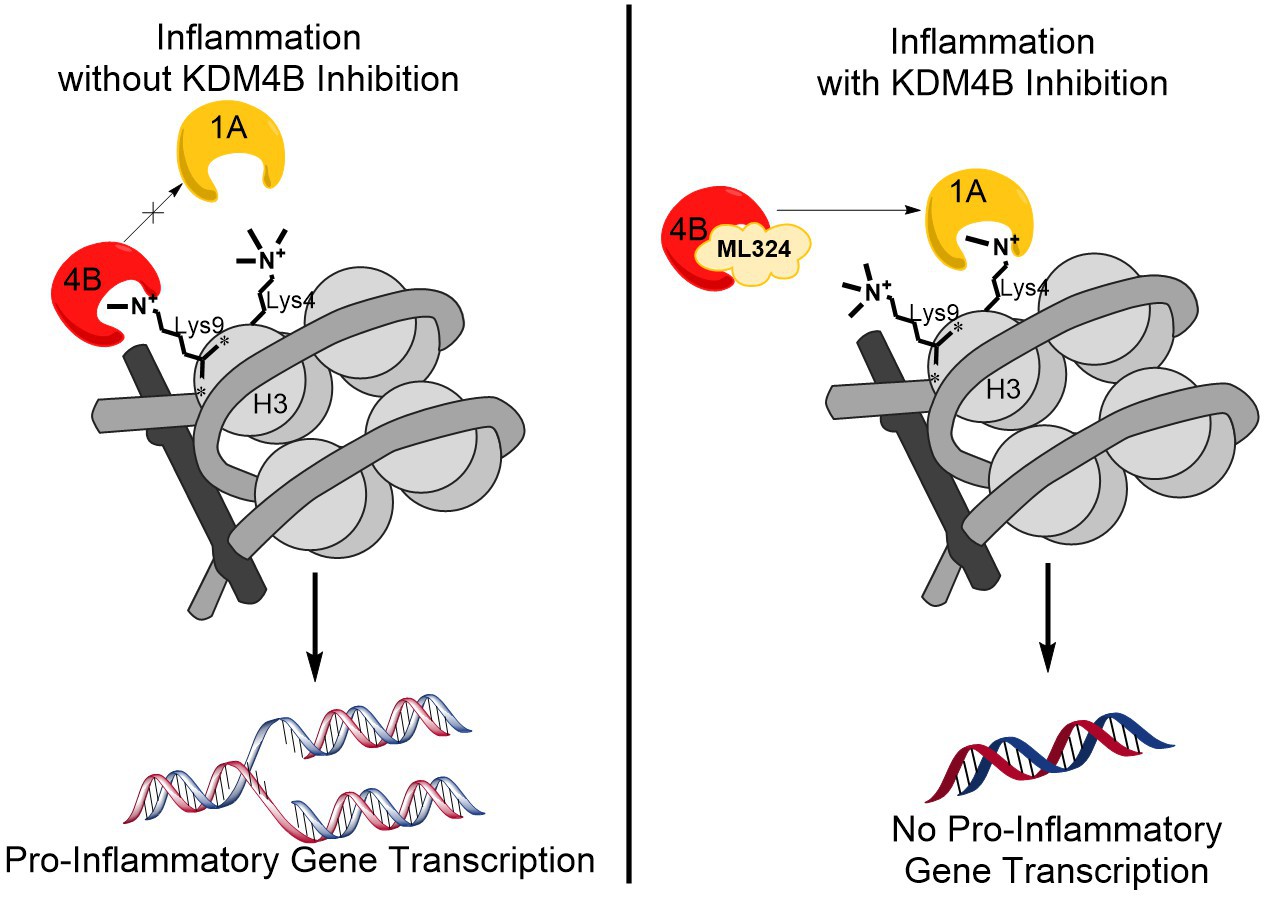
Results: KDM4B protein abundance was significantly higher in clinically diseased periodontal tissues compared to healthy controls as well as in areas of inflammatory infiltrate following live A.a injection. Pre-treatment of BMDMs with ML324 resulted in a significant reduction in A.a LPS induced pro-inflammatory cytokine transcription and translation in both males and females. This effect is modulated indirectly through increased KDM1A demethylation at H3K4. Novel, but commercially available, inhibitors for KDM4B have been identified in silico through computational chemical screens. A custom derivative library was generated and synthetic schemes were developed for synthesis of derivatives.
Conclusions: KDM4B has been identified as a promising new therapeutic target for treatment of PD through localized immunomodulation. In silico docking experiments have yielded several hit compounds allowing for creation of a custom derivative library. Synthesis of these therapeutics is ongoing, after which efficacy will be evaluated in vitro and in vivo.
Methods: KDM4B protein was quantified in both healthy and diseased human oral epithelium using immunohistochemistry. Twelve-week old C57BL/6 mice were evaluated in a 5-day calvarial PD model using live Aggregatibacter actinomycetemcomitans (A.a) injections. Tissue sections were stained using specific antibodies for TRAP, F4/80 and KDM4B. Bone-marrow-derived-macrophages (BMDMs) were isolated from 12-week old C57BL/6 male and female mice and pre-treated with the KDM4B inhibitor, ML324. Pre-treated BMDMs were challenged with Aggregatibacter actinomycetemcomitans lipopolysaccharide (A.a LPS) for 8 and 24 hours, after which RNA and supernatant proteins were collected for q-RT-PCR and ELISA analysis. RAW264.7 cells were subject to the same treatment and stained for methylation markers using the appropriate fluorescent antibodies. Computational docking experiments using the ZINC database were screened to identify potential KDM4B inhibitors for optimization. Statistical significance was determined using unpaired t-tests (p<0.05).
Results: KDM4B protein abundance was significantly higher in clinically diseased periodontal tissues compared to healthy controls as well as in areas of inflammatory infiltrate following live A.a injection. Pre-treatment of BMDMs with ML324 resulted in a significant reduction in A.a LPS induced pro-inflammatory cytokine transcription and translation in both males and females. This effect is modulated indirectly through increased KDM1A demethylation at H3K4. Novel, but commercially available, inhibitors for KDM4B have been identified in silico through computational chemical screens. A custom derivative library was generated and synthetic schemes were developed for synthesis of derivatives.
Conclusions: KDM4B has been identified as a promising new therapeutic target for treatment of PD through localized immunomodulation. In silico docking experiments have yielded several hit compounds allowing for creation of a custom derivative library. Synthesis of these therapeutics is ongoing, after which efficacy will be evaluated in vitro and in vivo.