IADR Abstract Archives
Age Comparisons in a RCT of Peri-implantitis Treatment With Minocycline-Microspheres
Objectives: The objective of the study is to evaluate the relation of age to the efficacy of Minocycline HCl 1mg Microspheres for the treatment of peri-implantitis used as an adjunct to mechanical debridement, when compared to debridement alone.
Methods: Two hundred eight (208) patients with implants diagnosed with peri-implantitis were enrolled in a multi-center trial and randomized to either mechanical debridement alone or mechanical debridement with the local administration of Minocycline HCl 1mg Microspheres. The primary outcome measure was probing depth reduction of qualifying implant sites at 6 months. Clinical assessments were performed at baseline, 3 and 6 months. Age subgroups (over fifty and below fifty) were analyzed for the primary outcome. Mixed model repeated measures was used to investigate the interaction of treatment by subgroup factor.
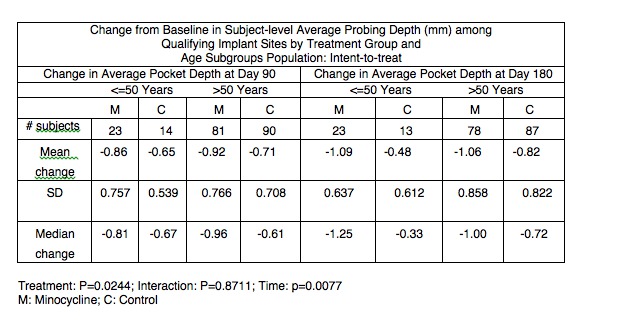
Results: In the total population, the subjects’ mean age was 61.4 years and 82.2% of the subjects were >50 years old. 50.5 % were male, 78.8% Caucasian, 16.8% Black, 2.9% Asian, 1% other, while 9.6% of subjects were smokers. The mean [SD] number of years since implant placement was 6.7 [4.05] years. Minocycline microspheres plus mechanical debridement provided more probing depth reduction than mechanical debridement alone in both subgroups (p=0.0244). Although Minocycline microspheres plus mechanical debridement provided more probing depth reduction than mechanical debridement alone in subjects ≤50 years than >50 years at 6 months, no evidence of interaction between the age subgroup and PD reduction was found (p=0.8711).
Conclusions: The adjunctive use of minocycline microspheres to the mechanical debridement of implants with peri-implantitis appears to provide an additional effect on the reduction of probing depths compared to mechanical debridement alone without any relation to the subjects’ age. Further studies including a bigger sample size of the young cohort are recommended.
Methods: Two hundred eight (208) patients with implants diagnosed with peri-implantitis were enrolled in a multi-center trial and randomized to either mechanical debridement alone or mechanical debridement with the local administration of Minocycline HCl 1mg Microspheres. The primary outcome measure was probing depth reduction of qualifying implant sites at 6 months. Clinical assessments were performed at baseline, 3 and 6 months. Age subgroups (over fifty and below fifty) were analyzed for the primary outcome. Mixed model repeated measures was used to investigate the interaction of treatment by subgroup factor.
Results: In the total population, the subjects’ mean age was 61.4 years and 82.2% of the subjects were >50 years old. 50.5 % were male, 78.8% Caucasian, 16.8% Black, 2.9% Asian, 1% other, while 9.6% of subjects were smokers. The mean [SD] number of years since implant placement was 6.7 [4.05] years. Minocycline microspheres plus mechanical debridement provided more probing depth reduction than mechanical debridement alone in both subgroups (p=0.0244). Although Minocycline microspheres plus mechanical debridement provided more probing depth reduction than mechanical debridement alone in subjects ≤50 years than >50 years at 6 months, no evidence of interaction between the age subgroup and PD reduction was found (p=0.8711).
Conclusions: The adjunctive use of minocycline microspheres to the mechanical debridement of implants with peri-implantitis appears to provide an additional effect on the reduction of probing depths compared to mechanical debridement alone without any relation to the subjects’ age. Further studies including a bigger sample size of the young cohort are recommended.