IADR Abstract Archives
High-velocity microsprays enhance interproximal oral biofilms detachment and dentifrice penetration
Objectives: Biofilms have a complex structure which can increase antimicrobials tolerance by limiting the diffusion of dentifrices inside the biofilm. Here, we assess Streptococcus mutans biofilm removal when exposed to various high-speed microsprays and the enhancement of dentifrice transport inside any remaining biofilm
Methods: Three-days-old S. mutans biofilm-colonized slides were placed inside a rectangular holder to simulate a 1-mm-interproximal space. Biofilms were exposed to high-velocity (vmax~50 m/s) water microsprays generated from two Philips dental devices, the AirFloss and AirFloss Pro which respectively produced a single 60 ms microspray or a triple 1 s microspray. Biofilm removal during the impact was recorded with a high-speed camera (HSC). Next, antimicrobial penetration was assessed by exposing biofilms to a 90° or 30° impact, firstly using a 1-µm tracer beads solution (109 beads/mL) and secondly, a 0.2% chlorexidine (CHX) or 0.085% cetylpyridinium chloride (CPC) solution. For comparison, diffusive transport (t=30 s) and a simulated mouth-washing (t=30 s, v=200 rpm) were performed. Confocal microscopy was used to determine number and relative bead penetration depth (PD) into the biofilm. For antimicrobials, the penetration depth was calculated from the resultant zone of killing detected by live/dead viability staining. Experiments were performed in triplicate.
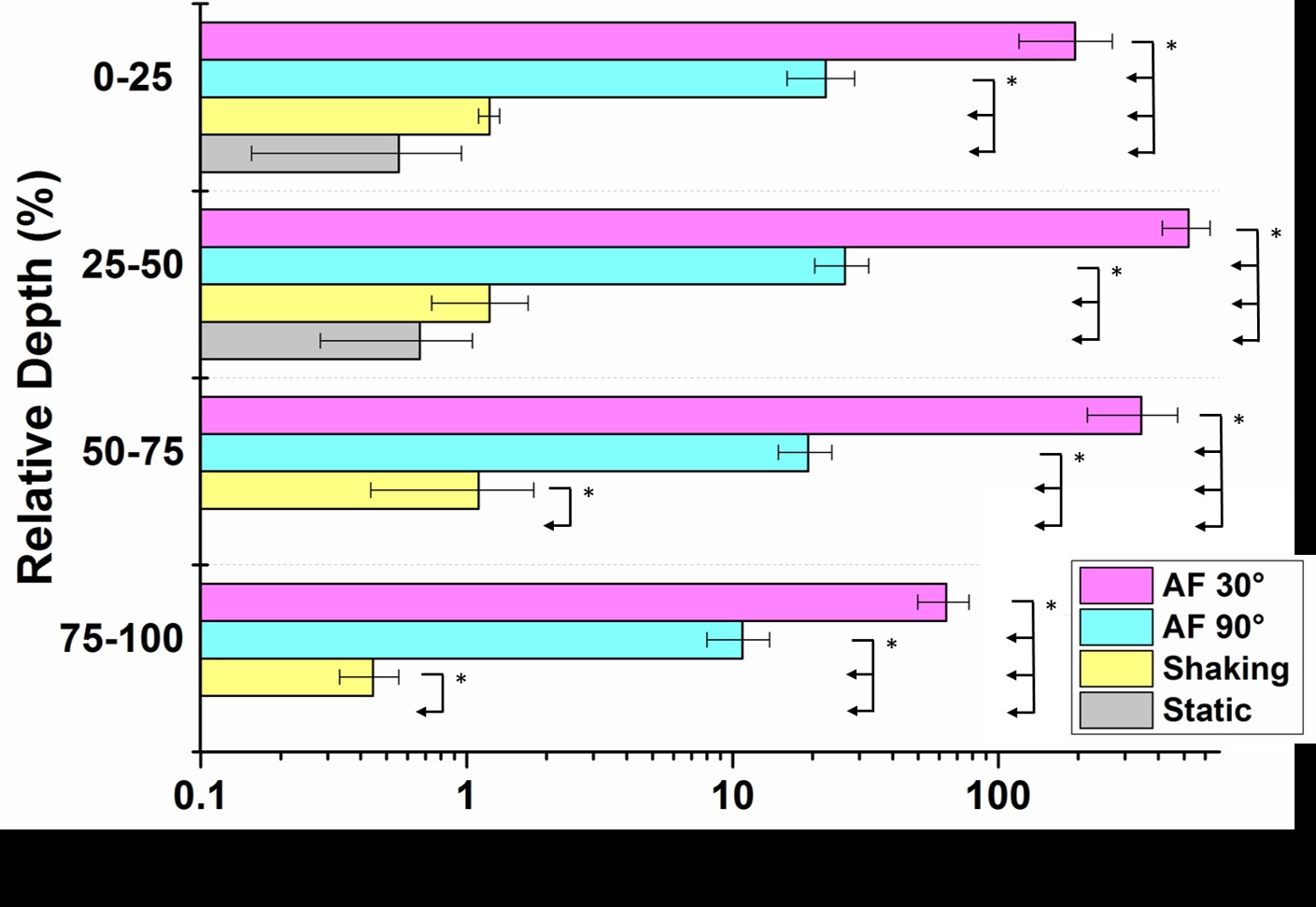
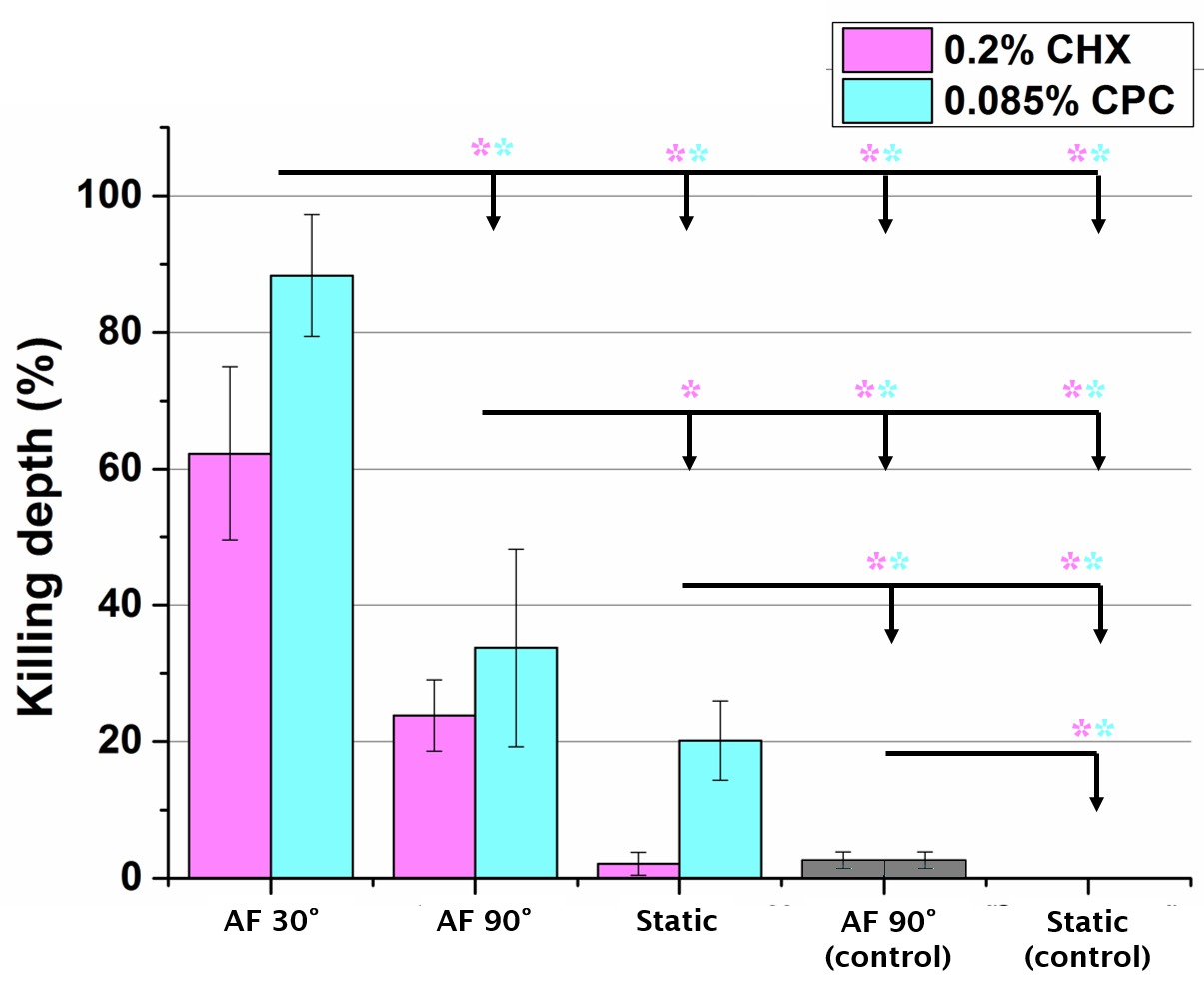
Results: The triple microspray removed twice as much biofilm (A=270.6±18.1 mm2) than the single microspray (130.1±13.1 mm2). Biofilm removal rate (u) decreased exponentially from u=1.9±0.32 m/s or was almost instantaneous (u=4.2±1.4 m/s) for the single and the triple microspray respectively. Single microspray shooting with an impact angle of 30° delivered significantly (p<0.05, n=3) more beads and yielded better antimicrobial penetration into the biofilm than a 90° impact (Fig1 and Fig2). However, both inclinations delivered significantly more beads and antimicrobials compared to a static or shaking conditions (Fig2).
Conclusions: Future work will incorporate antimicrobials and mouthrinses inside AirFloss microspray triple burst to to optimize biofilm removal and dentifrice delivery.
Methods: Three-days-old S. mutans biofilm-colonized slides were placed inside a rectangular holder to simulate a 1-mm-interproximal space. Biofilms were exposed to high-velocity (vmax~50 m/s) water microsprays generated from two Philips dental devices, the AirFloss and AirFloss Pro which respectively produced a single 60 ms microspray or a triple 1 s microspray. Biofilm removal during the impact was recorded with a high-speed camera (HSC). Next, antimicrobial penetration was assessed by exposing biofilms to a 90° or 30° impact, firstly using a 1-µm tracer beads solution (109 beads/mL) and secondly, a 0.2% chlorexidine (CHX) or 0.085% cetylpyridinium chloride (CPC) solution. For comparison, diffusive transport (t=30 s) and a simulated mouth-washing (t=30 s, v=200 rpm) were performed. Confocal microscopy was used to determine number and relative bead penetration depth (PD) into the biofilm. For antimicrobials, the penetration depth was calculated from the resultant zone of killing detected by live/dead viability staining. Experiments were performed in triplicate.
Results: The triple microspray removed twice as much biofilm (A=270.6±18.1 mm2) than the single microspray (130.1±13.1 mm2). Biofilm removal rate (u) decreased exponentially from u=1.9±0.32 m/s or was almost instantaneous (u=4.2±1.4 m/s) for the single and the triple microspray respectively. Single microspray shooting with an impact angle of 30° delivered significantly (p<0.05, n=3) more beads and yielded better antimicrobial penetration into the biofilm than a 90° impact (Fig1 and Fig2). However, both inclinations delivered significantly more beads and antimicrobials compared to a static or shaking conditions (Fig2).
Conclusions: Future work will incorporate antimicrobials and mouthrinses inside AirFloss microspray triple burst to to optimize biofilm removal and dentifrice delivery.