IADR Abstract Archives
Nanocatalytic Shelters Reversed Neutrophil-Macrophage Inflammatory Cascade in Chronic Inflammation
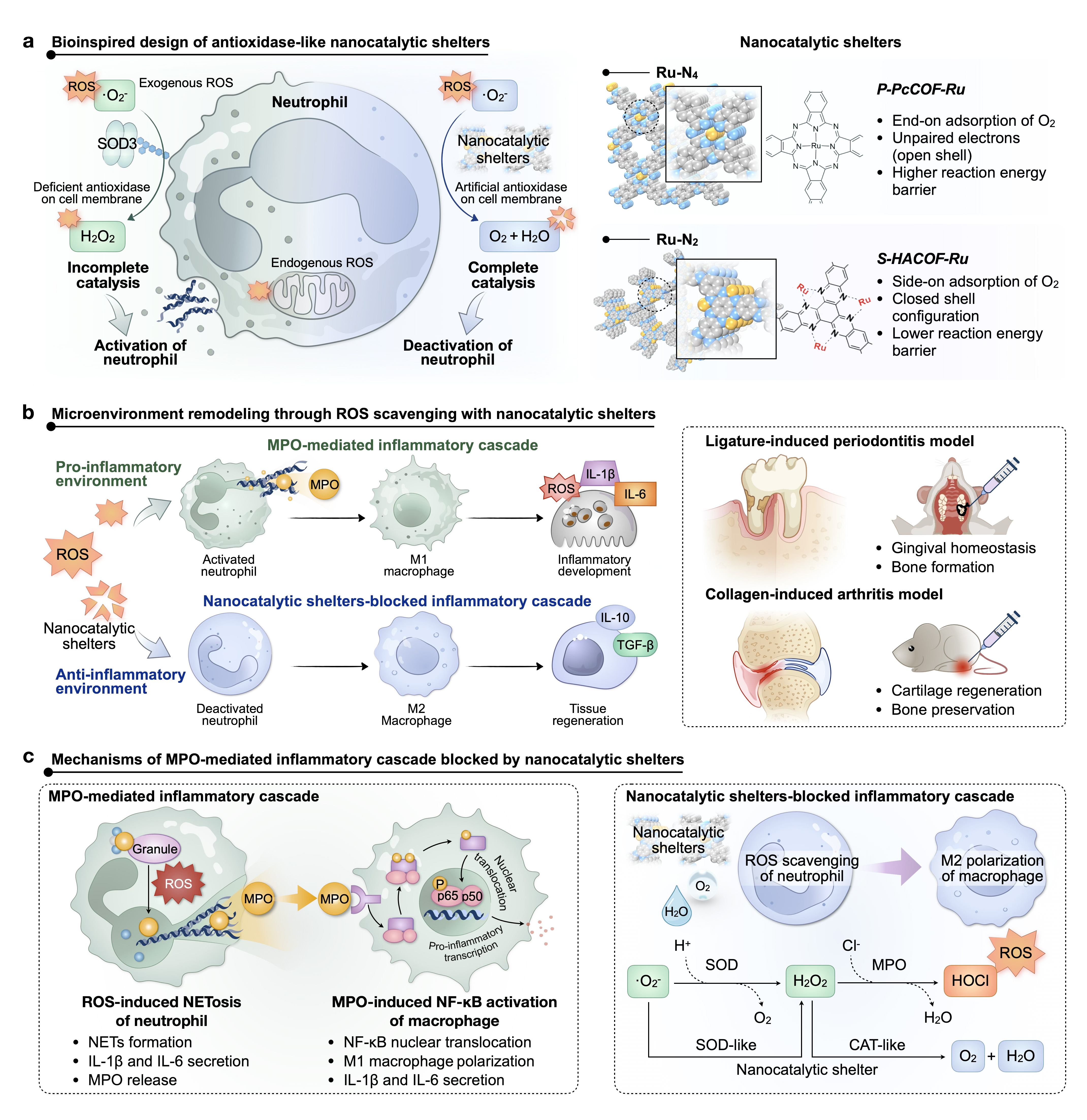
Objectives: Hyperactive neutrophils propagate chronic inflammation by engaging positive feedback loops and recruiting monocytes, leading to the overproduction of reactive oxygen species (ROS) and subsequent tissue damage. Targeting the ROS-driven neutrophil-macrophage inflammatory cascade, we propose bioinspired nanocatalytic shelters for reversing inflammatory cascade to treat chronic inflammatory diseases.
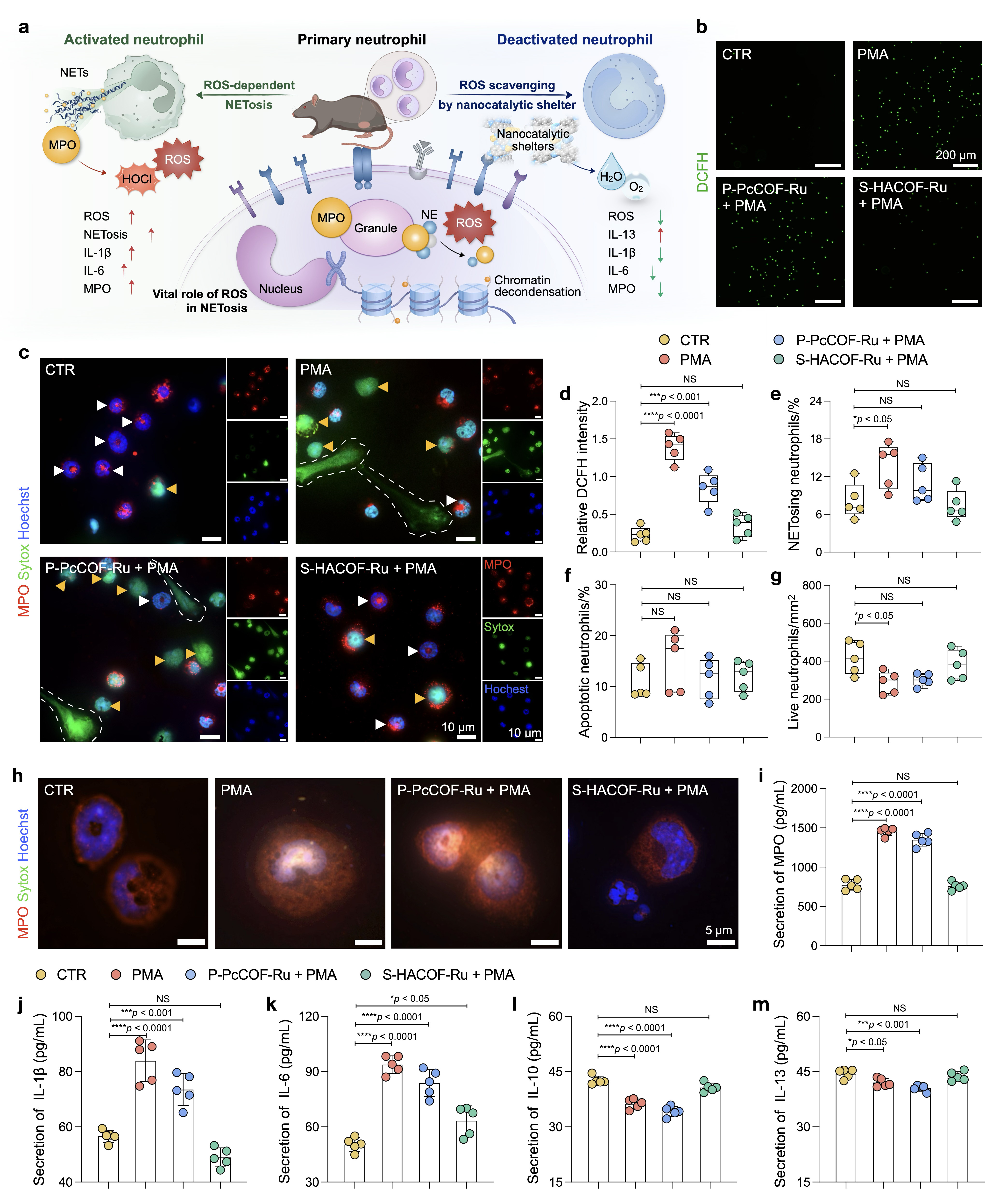
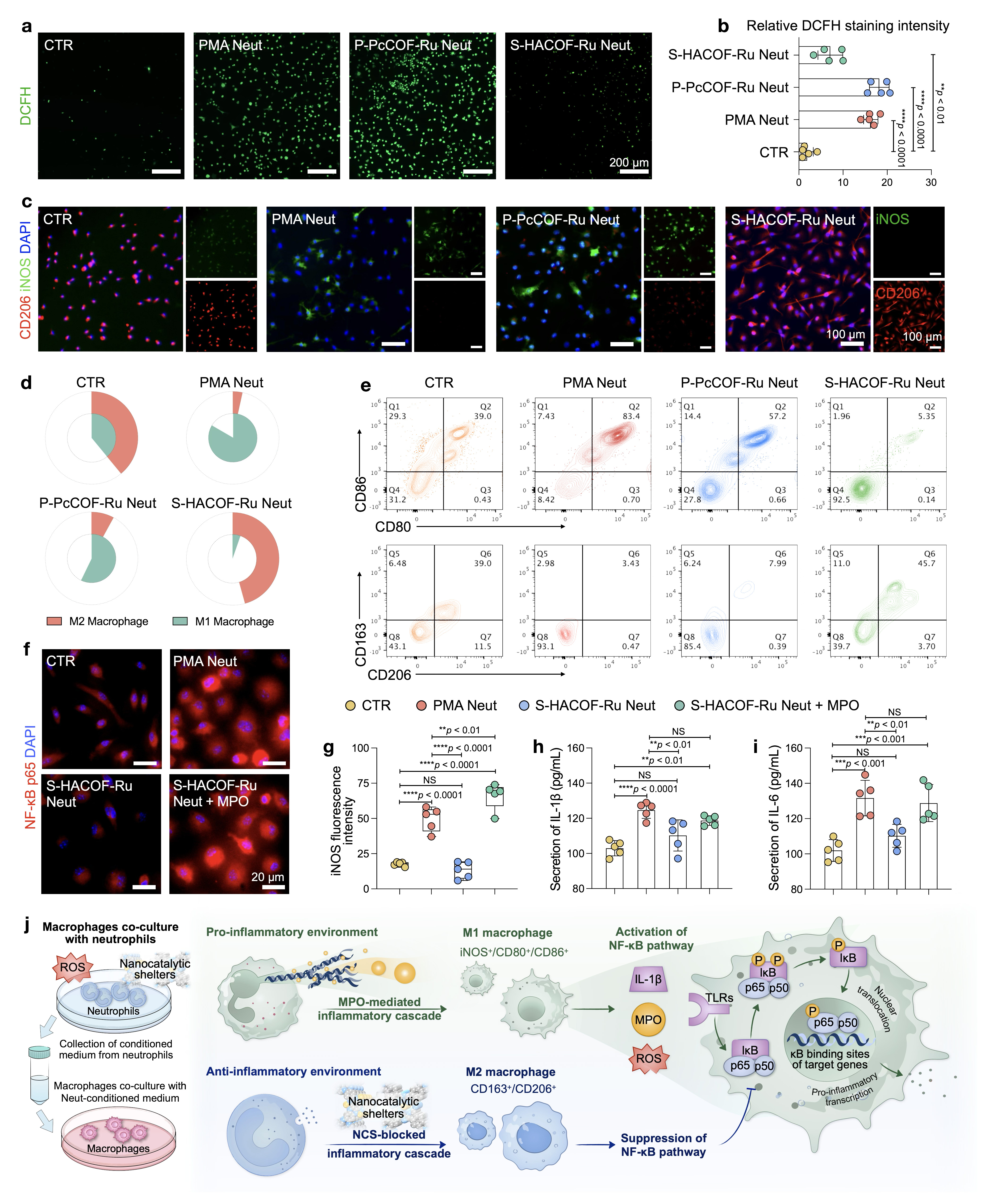
Methods: The covalent organic framework with Ru catalytic center in spatial configuration (S-HACOF-Ru) was synthesized, compared with the traditional phthalocyanine ruthenium polymer (P-PcCOF-Ru). Density functional theory (DFT) calculations elucidated the catalytic mechanisms. The nanocatalytic shelters were then applied to neutrophils to assess neutrophil extracellular traps (NETs), myeloperoxidase (MPO) release, and other immune functions. The mechanism of ROS-mediated disruption of inflammatory cascades was further investigated in the neutrophil-macrophage co-culture system. In vivo studies of periodontitis and rheumatoid arthritis (RA), integrating imaging, histological analysis, and transcriptomic sequencing, highlighted the role of nanocatalytic shelters in restoring immune homeostasis.
Results: A novel ruthenium single-atom-coordinated nitrogen-rich structure is characterized in S-HACOF-Ru, which displays superior biocatalytic activities for broad-spectrum ROS elimination. S-HACOF-Ru can simultaneously inhibit NETs and MPO. Through the prevention of MPO-induced NF-κB activation and subsequent inhibition of M1 macrophage polarization, thereby blocking the inflammatory cascade. By exerting significant therapeutic potential, the S-HACOF-Ru efficiently restores alveolar bone loss in the periodontal tissue and reverses cartilage damage in the ankle joint cavity.
Conclusions: The nanocatalytic shelters effectively disrupt the neutrophil-macrophage inflammatory cascade by inhibiting NETosis and MPO release, reversing macrophage polarization, which is consistent with the alteration of inflammatory progression to tissue repair in periodontitis and RA treatment, offering a promising avenue for antioxidase-like materials to address a broad range of chronic inflammation.
Methods: The covalent organic framework with Ru catalytic center in spatial configuration (S-HACOF-Ru) was synthesized, compared with the traditional phthalocyanine ruthenium polymer (P-PcCOF-Ru). Density functional theory (DFT) calculations elucidated the catalytic mechanisms. The nanocatalytic shelters were then applied to neutrophils to assess neutrophil extracellular traps (NETs), myeloperoxidase (MPO) release, and other immune functions. The mechanism of ROS-mediated disruption of inflammatory cascades was further investigated in the neutrophil-macrophage co-culture system. In vivo studies of periodontitis and rheumatoid arthritis (RA), integrating imaging, histological analysis, and transcriptomic sequencing, highlighted the role of nanocatalytic shelters in restoring immune homeostasis.
Results: A novel ruthenium single-atom-coordinated nitrogen-rich structure is characterized in S-HACOF-Ru, which displays superior biocatalytic activities for broad-spectrum ROS elimination. S-HACOF-Ru can simultaneously inhibit NETs and MPO. Through the prevention of MPO-induced NF-κB activation and subsequent inhibition of M1 macrophage polarization, thereby blocking the inflammatory cascade. By exerting significant therapeutic potential, the S-HACOF-Ru efficiently restores alveolar bone loss in the periodontal tissue and reverses cartilage damage in the ankle joint cavity.
Conclusions: The nanocatalytic shelters effectively disrupt the neutrophil-macrophage inflammatory cascade by inhibiting NETosis and MPO release, reversing macrophage polarization, which is consistent with the alteration of inflammatory progression to tissue repair in periodontitis and RA treatment, offering a promising avenue for antioxidase-like materials to address a broad range of chronic inflammation.