IADR Abstract Archives
Hypoxia-MSC-Derived Apoptotic Extracellular Vesicles in 3D-Printed Scaffolds Promote Osteochondral Regeneration
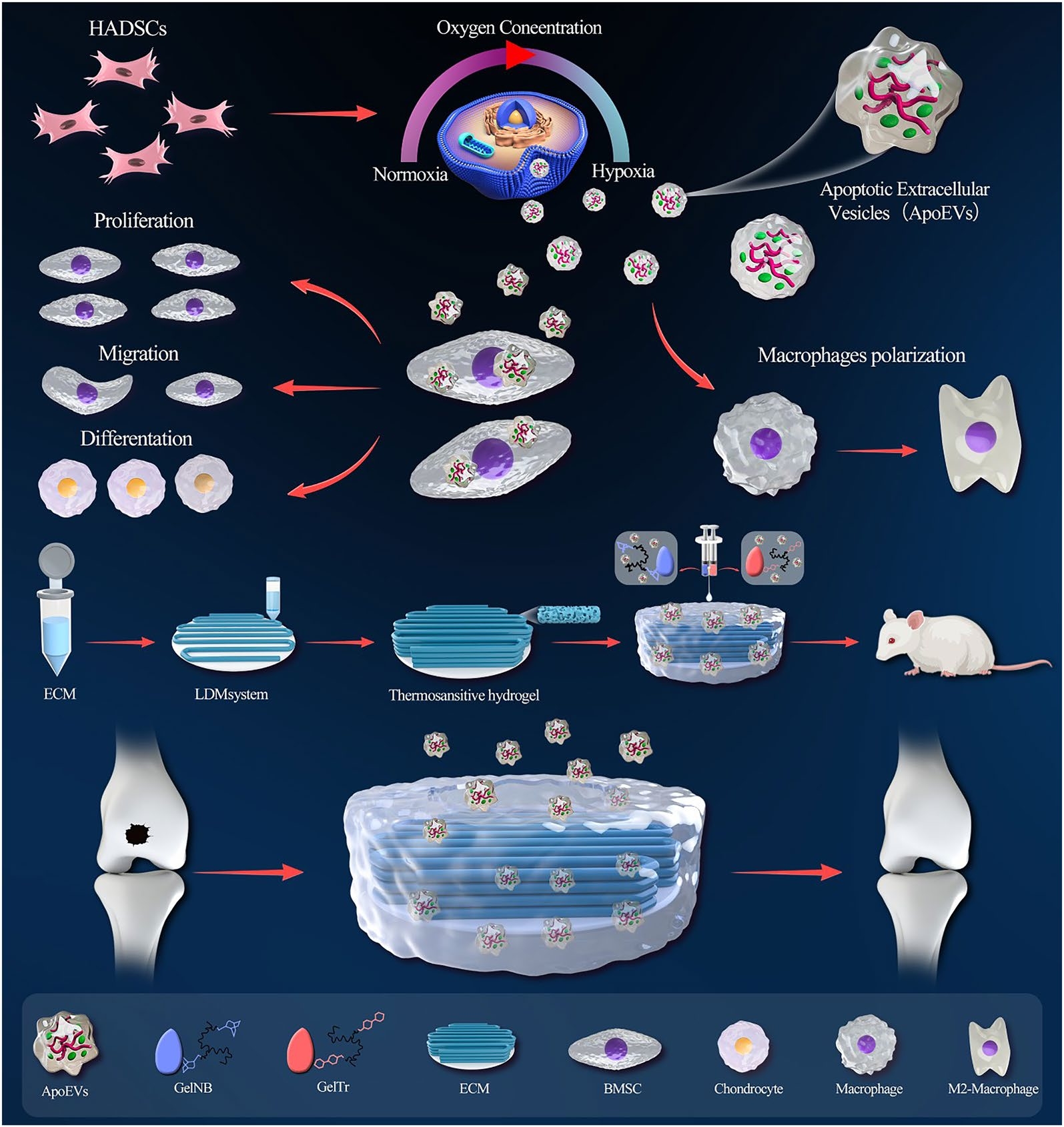
Objectives: Due to the unique structure, articular cartilage cannot undergo self-repair after injury, causing a difficult problem in the field of sports medicine. The objective of this study is to explore whether the apoptotic extracellular vesicles (ApoEVs) derived from MSCs cultured under hypoxic conditions (hypoxic apoptotic EVs [H-ApoEVs]) exert greater effects on cartilage repair than those cultured under normoxic conditions. Moreover, a carrier is developed, and the delivery efficiency of H-ApoEVs into the joint cavity for osteochondral regeneration is investigated.
Methods: First, MSCs were transplanted into a rat osteochondral defect model, and their fate was observed. Then, human adipose-derived MSCs were cultured under normoxic and hypoxic conditions, following the induction of apoptosis, ApoEVs extraction, BMSC proliferation and migration, chondrogenic differentiation, and macrophage polarization. Next, miRNA sequencing and proteomics analysis of H-ApoEVs and ApoEVs was performed, and mRNA sequencing on BMSCs was conducted after treating with different ApoEVs. Finally, a carrier composed of a click-chemistry crosslinking gelatin matrix and 3D-printed cartilage extracellular matrix scaffold was developed to deliver H-ApoEVs into the rat osteochondral defect model.
Results: In vitro cell proliferation and migration experiments demonstrated that H-ApoEVs enhanced stem cell proliferation, migration, and bone marrow-derived macrophages (BMDMs) M2 polarization compared to ApoEVs. The 3D-printed ECM scaffold provided mechanical support and created a microenvironment that was conducive to cartilage regeneration, and the H-ApoEVs further enhanced the regenerative capacity of endogenous stem cells and the immunomodulatory microenvironment of the joint cavity; thus, this approach significantly promoted cartilage regeneration.
Conclusions: In conclusion, this study confirmed that the strategy to complex hypoxia-MSC-derived ApoEVs in a click-chemistry crosslinking gelatin matrix / 3D-printed cartilage ECM scaffold represents a promising approach to promote cartilage regeneration.
Methods: First, MSCs were transplanted into a rat osteochondral defect model, and their fate was observed. Then, human adipose-derived MSCs were cultured under normoxic and hypoxic conditions, following the induction of apoptosis, ApoEVs extraction, BMSC proliferation and migration, chondrogenic differentiation, and macrophage polarization. Next, miRNA sequencing and proteomics analysis of H-ApoEVs and ApoEVs was performed, and mRNA sequencing on BMSCs was conducted after treating with different ApoEVs. Finally, a carrier composed of a click-chemistry crosslinking gelatin matrix and 3D-printed cartilage extracellular matrix scaffold was developed to deliver H-ApoEVs into the rat osteochondral defect model.
Results: In vitro cell proliferation and migration experiments demonstrated that H-ApoEVs enhanced stem cell proliferation, migration, and bone marrow-derived macrophages (BMDMs) M2 polarization compared to ApoEVs. The 3D-printed ECM scaffold provided mechanical support and created a microenvironment that was conducive to cartilage regeneration, and the H-ApoEVs further enhanced the regenerative capacity of endogenous stem cells and the immunomodulatory microenvironment of the joint cavity; thus, this approach significantly promoted cartilage regeneration.
Conclusions: In conclusion, this study confirmed that the strategy to complex hypoxia-MSC-derived ApoEVs in a click-chemistry crosslinking gelatin matrix / 3D-printed cartilage ECM scaffold represents a promising approach to promote cartilage regeneration.