IADR Abstract Archives
ATF2 Promotes Osteoblast Differentiation Through Phospharylation-Dependent Activation of Runx2
Objectives: Osteoblast differentiation is spatiotemporally regulated by transcription factors. Our previous work revealed a correlation between p-ATF2 and increased chromatin accessibility of regions near mineralization-related genes. Nevertheless, the detailed mechanism by which ATF2 initiates its downstream transcription factor cascade and regulates osteoblastic differentiation remains elusive.
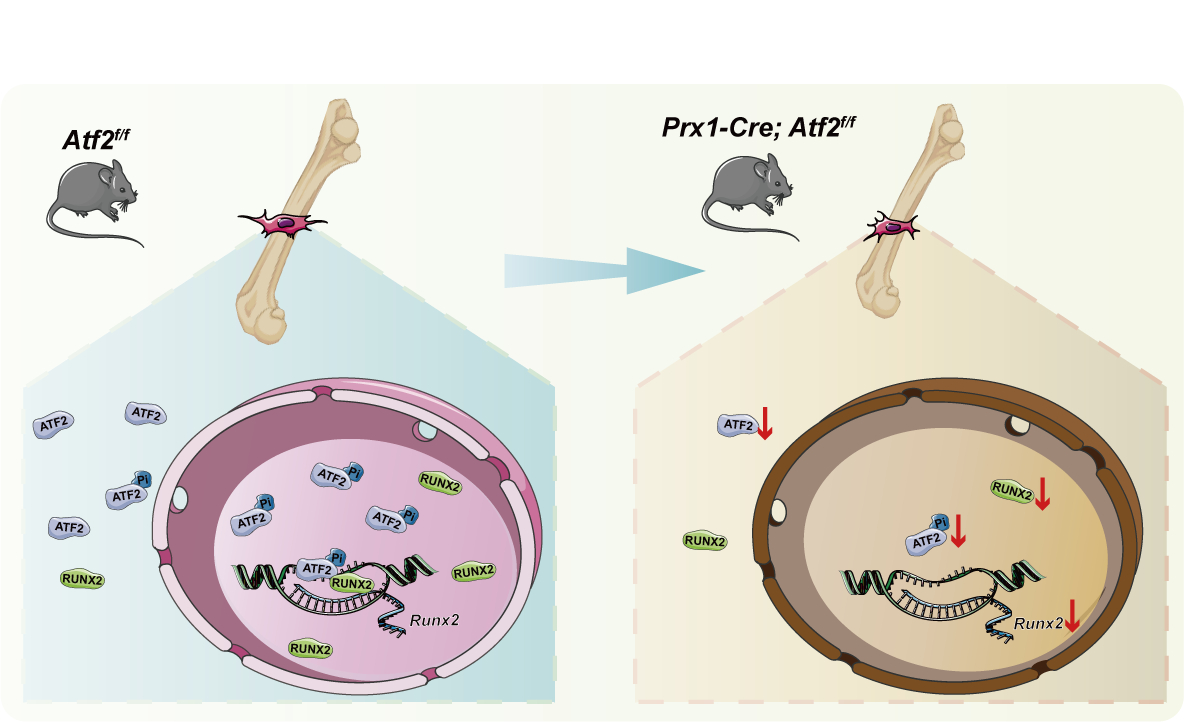
Methods: (i) The expression of ATF2 was detected during bone development. (ii) Prx1-Cre; Atf2f/f mice were constructed to knockout Atf2 in mesodermal mesenchyme. (iii) The target genes and pathways regulated by ATF2 following Atf2 deletion were elucidated through RNA-seq and GO analysis. (iv) The effects on osteoblast differentiation were also detected following overexpression of p-ATF2. (v) The fluorescence double staining was used to detect the colocalization of p-ATF2 and RUNX2. The co-IP and PLA assays were employed to investigate the interaction between p-ATF2 and RUNX2. (vi) CUT&Tag-qPCR were used to determine whether p-ATF2 binds to the transcriptional regulatory regions, a dual luciferase assay was conducted to verify the transcriptional activity of p-ATF2.
Results: (i) ATF2 was highly expressed in hypertrophic chondrocytes and preosteoblasts but weakly expressed in proliferating chondrocytes. (ii) The micro-CT results showed underdeveloped long bones and decreased bone density in the ATF2-cKO mice. H&E staining confirmed that the trabecular bone number and the width of cortical bone were reduced in ATF2-cKO. (iii) RNA-seq results showed that downregulated genes were enriched in mineralization-related GO terms. (iv) The expression levels of p-ATF2 and differentiation marker genes only increased in the p-ATF2 group. (v) p-ATF2 and RUNX2 had similar nuclear localization and expression trends after differentiation induction. p-ATF2 interacted and colocalized with RUNX2. (vi) CUT&Tag-qPCR experiments demonstrated p-ATF2 binding to the chromatin region of Runx2 gene, the dual luciferase assay confirmed transcriptional activity of p-ATF2 on the cis-acting element of Runx2.
Conclusions: During osteoblast differentiation, ATF2 promotes osteoblast differentiation and bone development by transcriptionally activating differentiation-related genes.
Methods: (i) The expression of ATF2 was detected during bone development. (ii) Prx1-Cre; Atf2f/f mice were constructed to knockout Atf2 in mesodermal mesenchyme. (iii) The target genes and pathways regulated by ATF2 following Atf2 deletion were elucidated through RNA-seq and GO analysis. (iv) The effects on osteoblast differentiation were also detected following overexpression of p-ATF2. (v) The fluorescence double staining was used to detect the colocalization of p-ATF2 and RUNX2. The co-IP and PLA assays were employed to investigate the interaction between p-ATF2 and RUNX2. (vi) CUT&Tag-qPCR were used to determine whether p-ATF2 binds to the transcriptional regulatory regions, a dual luciferase assay was conducted to verify the transcriptional activity of p-ATF2.
Results: (i) ATF2 was highly expressed in hypertrophic chondrocytes and preosteoblasts but weakly expressed in proliferating chondrocytes. (ii) The micro-CT results showed underdeveloped long bones and decreased bone density in the ATF2-cKO mice. H&E staining confirmed that the trabecular bone number and the width of cortical bone were reduced in ATF2-cKO. (iii) RNA-seq results showed that downregulated genes were enriched in mineralization-related GO terms. (iv) The expression levels of p-ATF2 and differentiation marker genes only increased in the p-ATF2 group. (v) p-ATF2 and RUNX2 had similar nuclear localization and expression trends after differentiation induction. p-ATF2 interacted and colocalized with RUNX2. (vi) CUT&Tag-qPCR experiments demonstrated p-ATF2 binding to the chromatin region of Runx2 gene, the dual luciferase assay confirmed transcriptional activity of p-ATF2 on the cis-acting element of Runx2.
Conclusions: During osteoblast differentiation, ATF2 promotes osteoblast differentiation and bone development by transcriptionally activating differentiation-related genes.