IADR Abstract Archives
Efficacy of MucoPEG in Relieving Symptoms of Xerostomia
Objectives: The primary objective of this study was to evaluate the effect of MucoPEGTM in relieving xerostomia, relative to Biotène® Dry Mouth Gentle Oral Rinse. Effects of both oral rinse products were assessed using the Visual Analogue Scale (VAS) and Dry Mouth Relief Questionnaire.
Methods: Forty-two xerostomic subjects were enrolled at 2 sites using an open-label randomized cross-over design. Subjects were randomized into 2 groups, each using Biotene and MucoPEG for 2 weeks in opposing order, with a one-week washout period. Subjects completed VAS, dry mouth relief questionnaires, dry mouth Product Performance and Attributes Questionnaire (PPAQ), and Post-Product Use Questionnaire (PPUQ) at multiple timepoints to assess their subjective feelings of oral dryness. This study was approved by Tufts University Health Sciences IRB (Tufts) and Advarra CIRB (Colorado).
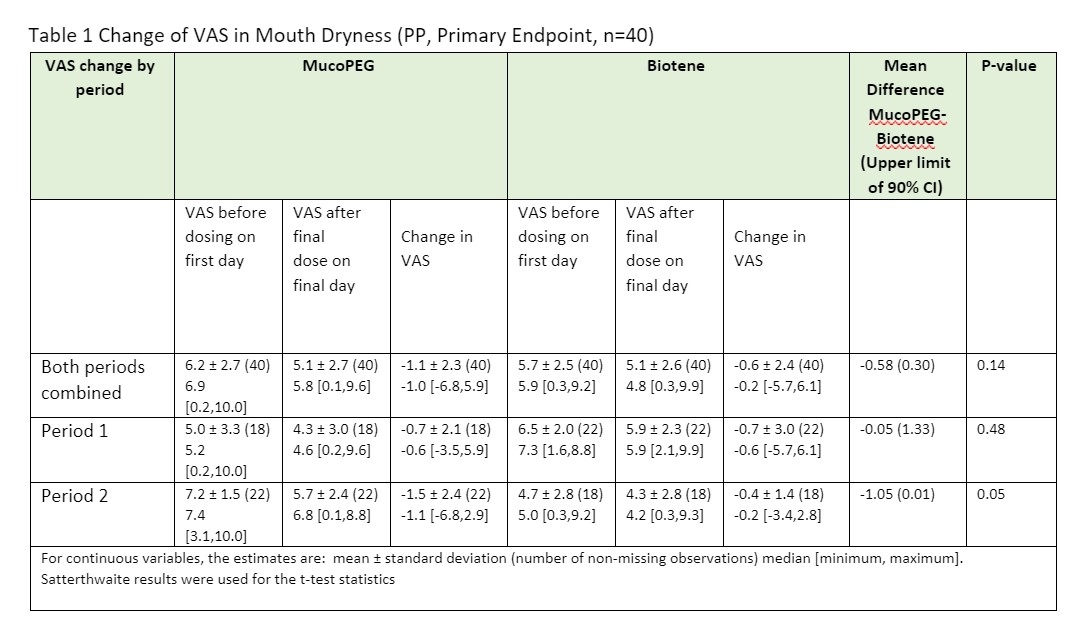
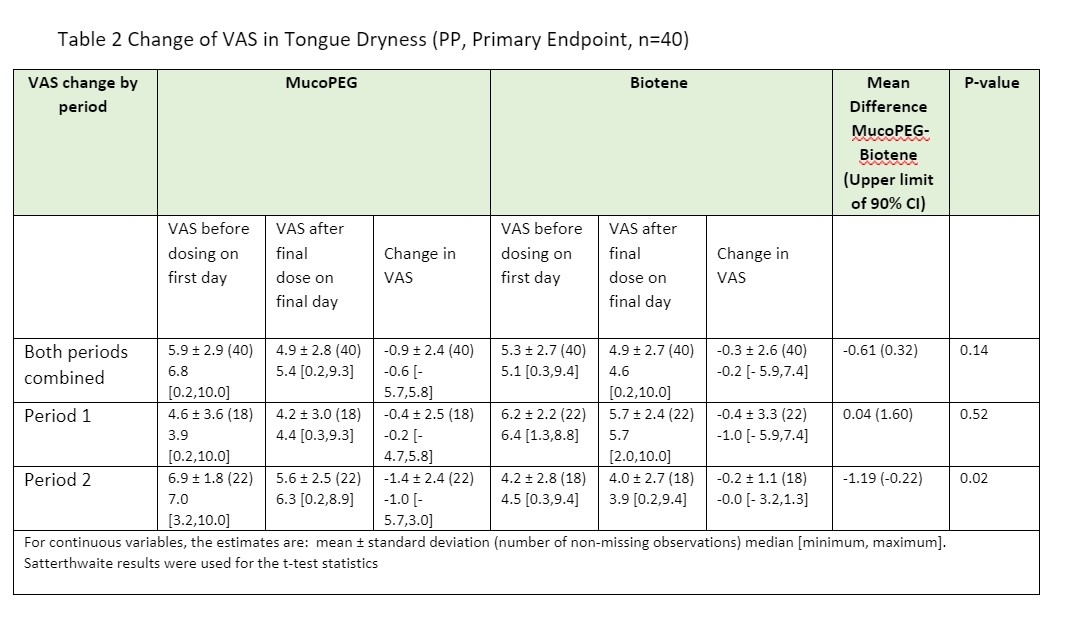
Results: MucoPEG showed greater improvement in symptoms of mouth dryness compared to Biotene (p-value of VAS; for Mouth dryness: 0.05, Tongue dryness: 0.02) in subjects under Period 2, and greater improvement in symptoms (p-value of VAS; for Mouth dryness: 0.14, Tongue dryness: 0.14) for both periods combined. (Tables 1 and 2). MucoPEG demonstrated non-inferiority to Biotene but failed to demonstrate superior efficacy due to lacking enough power to confirm statistical significance.
Conclusions: Xerostomic patients need more effective and long-lasting products to relieve their symptoms and reduce their risk of oral diseases. MucoPEG showed effectiveness at improving some symptoms of xerostomia in this xerostomic population. Larger studies are needed to attain more conclusive results. This study was funded by SunBio, Inc.
Methods: Forty-two xerostomic subjects were enrolled at 2 sites using an open-label randomized cross-over design. Subjects were randomized into 2 groups, each using Biotene and MucoPEG for 2 weeks in opposing order, with a one-week washout period. Subjects completed VAS, dry mouth relief questionnaires, dry mouth Product Performance and Attributes Questionnaire (PPAQ), and Post-Product Use Questionnaire (PPUQ) at multiple timepoints to assess their subjective feelings of oral dryness. This study was approved by Tufts University Health Sciences IRB (Tufts) and Advarra CIRB (Colorado).
Results: MucoPEG showed greater improvement in symptoms of mouth dryness compared to Biotene (p-value of VAS; for Mouth dryness: 0.05, Tongue dryness: 0.02) in subjects under Period 2, and greater improvement in symptoms (p-value of VAS; for Mouth dryness: 0.14, Tongue dryness: 0.14) for both periods combined. (Tables 1 and 2). MucoPEG demonstrated non-inferiority to Biotene but failed to demonstrate superior efficacy due to lacking enough power to confirm statistical significance.
Conclusions: Xerostomic patients need more effective and long-lasting products to relieve their symptoms and reduce their risk of oral diseases. MucoPEG showed effectiveness at improving some symptoms of xerostomia in this xerostomic population. Larger studies are needed to attain more conclusive results. This study was funded by SunBio, Inc.