IADR Abstract Archives
Hydrophobic Binding Ability of Statherin on Hydroxyapatite (HAP)
Objectives: Statherin, one component of acquired enamel pellicle formed by selective adsorption of salivary proteins on teeth, could capture calcium ions which is vital for the calcium homeostasis and integrity of teeth. Studies suggest negative charge density and helical conformation at N-terminal of it are critical for its binding with HAP. However, hydrophobic interaction between statherin and HAP are poorly understood. Thus, comparison binding ability of statherin on HAP and hydrophobic surface was conducted.
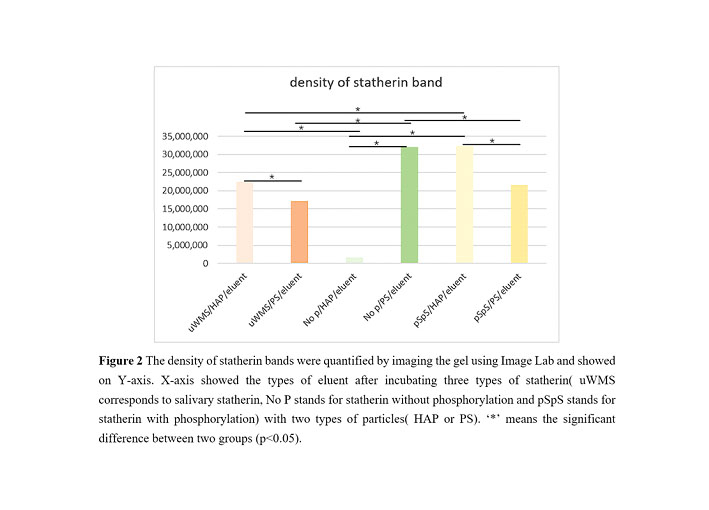
Methods: HAP and polystyrene particles, PS particles, with equal area were incubated with 0.5ml unstimulated whole mouth saliva (uWMS) or 40ug/ml synthetic statherin (+/-phosphorylation). After incubation, the mixtures were centrifuged and supernatants were removed. 100ul EDTA and 100ul TBST (tris-buffered saline Tween-20) were added to HAP and PS pellets respectively to elute the proteins bound after washing. The supernatants were collected after centrifuging followed by electrophoretic analysis on density of statherin bands. Two-way ANOVA and Tukey’s test were performed between the two binding surfaces and three types of statherin.
Results: Two-way ANOVA revealed that both the statherin types, binding surfaces and their interaction significantly influenced binding amount on particles (p<0.05). On HAP, synthetic phosphorylated statherin had statistically higher binding amount than salivary statherin followed by non-phosphorylated synthetic statherin (p<0.05). On PS particles, non-phosphorylated synthetic statherin achieved higher binding amount compared to the rest two types of statherin (p<0.05). Between different types of statherin, both salivary statherin and synthetic phosphorylated statherin obtained higher binding amount on HAP than PS particles whose amount had accounted for about 70% or above that amount on HAP (p<0.05).
Conclusions: Hydrophobic interaction seems to account for the main source of binding of statherin from saliva with HAP and phosphorylation has no impact on this. Some factors may exists in saliva that inhibits the binding of statherin on HAP.
Methods: HAP and polystyrene particles, PS particles, with equal area were incubated with 0.5ml unstimulated whole mouth saliva (uWMS) or 40ug/ml synthetic statherin (+/-phosphorylation). After incubation, the mixtures were centrifuged and supernatants were removed. 100ul EDTA and 100ul TBST (tris-buffered saline Tween-20) were added to HAP and PS pellets respectively to elute the proteins bound after washing. The supernatants were collected after centrifuging followed by electrophoretic analysis on density of statherin bands. Two-way ANOVA and Tukey’s test were performed between the two binding surfaces and three types of statherin.
Results: Two-way ANOVA revealed that both the statherin types, binding surfaces and their interaction significantly influenced binding amount on particles (p<0.05). On HAP, synthetic phosphorylated statherin had statistically higher binding amount than salivary statherin followed by non-phosphorylated synthetic statherin (p<0.05). On PS particles, non-phosphorylated synthetic statherin achieved higher binding amount compared to the rest two types of statherin (p<0.05). Between different types of statherin, both salivary statherin and synthetic phosphorylated statherin obtained higher binding amount on HAP than PS particles whose amount had accounted for about 70% or above that amount on HAP (p<0.05).
Conclusions: Hydrophobic interaction seems to account for the main source of binding of statherin from saliva with HAP and phosphorylation has no impact on this. Some factors may exists in saliva that inhibits the binding of statherin on HAP.