IADR Abstract Archives
Salivary Levels of Hydrogen Peroxide After use of Whitening Product
Objectives: To determine the hydrogen peroxide (HP) content in patients’ saliva at different time points after application of the Access Dental Lab whitening product, Bright On™.
Methods: A pre-treatment unstimulated saliva sample and teeth shade were taken from 15 patients. The test product was applied to 6 front teeth (top and bottom) using a customized whitening pen applicator and the gel was left on for 30 minutes. Saliva samples during treatment were collected at the following time intervals: 1, 5, 10, and 30 minutes. During treatment, subjects were asked to pool the saliva in their mouth for 1 minute and their spit was collected using disposable pipettes. An additional saliva sample was collected after the bleaching product was washed of the teeth. In addition, a post-treatment shade was taken using a Vita Shade Guide. The amount of HP in the saliva samples were measured using a sensitive fluorometric assay kit (MAK165, Sigma Aldrich) following the manufacturer’s protocol. A standard curve was built for each assay and prepared from a 3% HP solution. Standards and samples were spotted, in duplicate, in a 96-well plate and the fluorescence measured using a Fluoroskan Ascent plate reader. The pH of the whitening product was measured using two methods of extraction into distilled water and vortex (method 1) as well as extraction into distilled water and homogenization using an IKA Ultra Turrax (method 2).
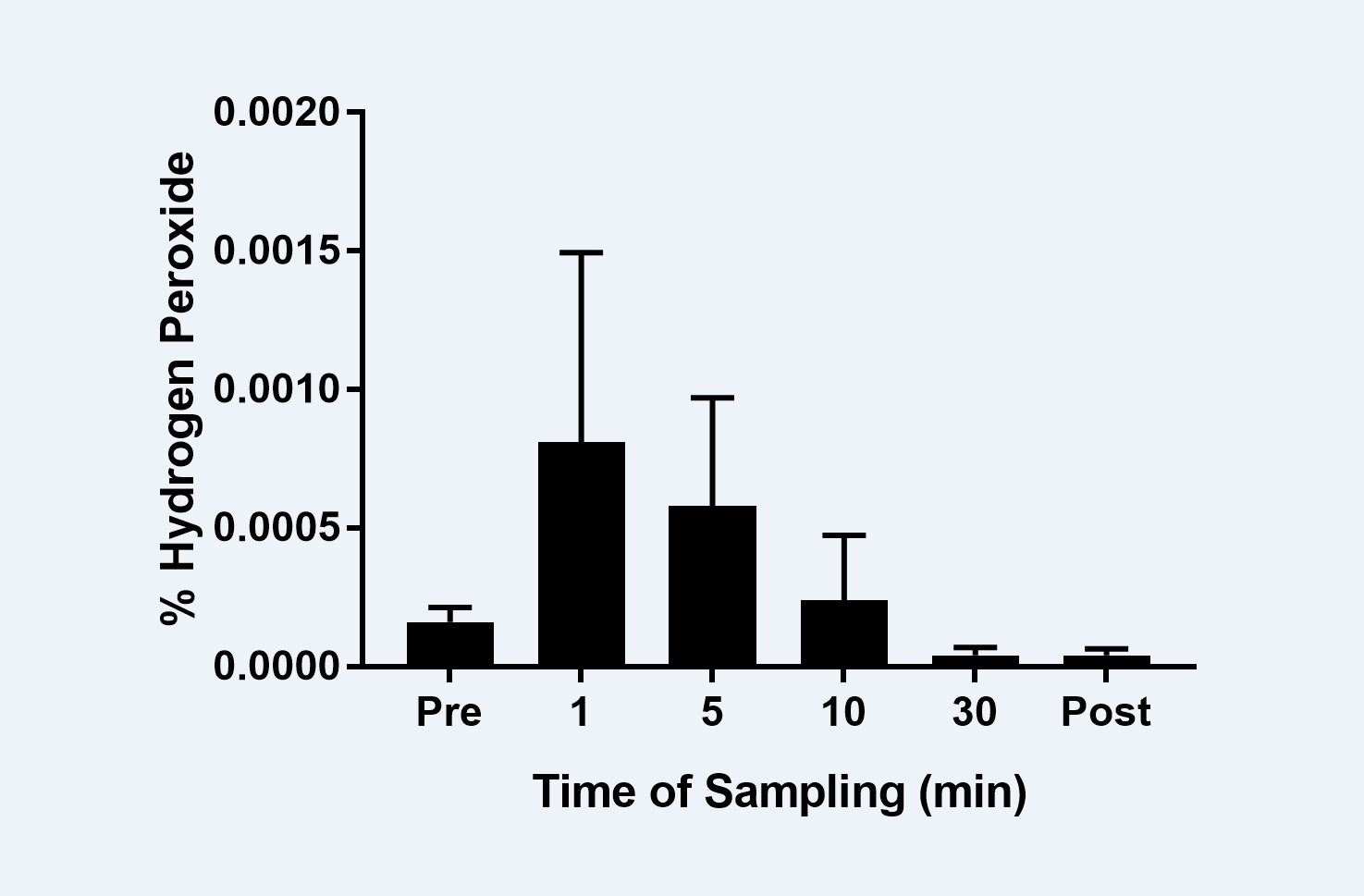
Results: The pH of the whitening product measured using method 1 was 5.69 ± 0.08, while method 2 yielded a pH value of 5.80 ± 0.16. As shown in Figure 1, the amount of HP in the saliva samples at all time points was negligible and well below the 3% threshold.
Conclusions: The levels of HP released from the withering product were consistently below the 3% safety threshold.
Methods: A pre-treatment unstimulated saliva sample and teeth shade were taken from 15 patients. The test product was applied to 6 front teeth (top and bottom) using a customized whitening pen applicator and the gel was left on for 30 minutes. Saliva samples during treatment were collected at the following time intervals: 1, 5, 10, and 30 minutes. During treatment, subjects were asked to pool the saliva in their mouth for 1 minute and their spit was collected using disposable pipettes. An additional saliva sample was collected after the bleaching product was washed of the teeth. In addition, a post-treatment shade was taken using a Vita Shade Guide. The amount of HP in the saliva samples were measured using a sensitive fluorometric assay kit (MAK165, Sigma Aldrich) following the manufacturer’s protocol. A standard curve was built for each assay and prepared from a 3% HP solution. Standards and samples were spotted, in duplicate, in a 96-well plate and the fluorescence measured using a Fluoroskan Ascent plate reader. The pH of the whitening product was measured using two methods of extraction into distilled water and vortex (method 1) as well as extraction into distilled water and homogenization using an IKA Ultra Turrax (method 2).
Results: The pH of the whitening product measured using method 1 was 5.69 ± 0.08, while method 2 yielded a pH value of 5.80 ± 0.16. As shown in Figure 1, the amount of HP in the saliva samples at all time points was negligible and well below the 3% threshold.
Conclusions: The levels of HP released from the withering product were consistently below the 3% safety threshold.