IADR Abstract Archives
Comparison of Flexural Strength of Bioactive-Glass Composites with Commercial Composites
Objectives: Composite restorations comprise most of current direct restorations and are manufactured using different filler and monomer configurations. Bioactive glass (BAG) has been shown to be biocompatible, have antimicrobial properties, release ions and stimulate apatite formation. Developing a BAG-filled composite with comparable mechanical and physical properties to composites that are currently available might increase the clinical success of direct restorations. This study measured and compared the flexural strength of 12 novel BAG-containing resin composites with 4 commercially available ones.
Methods: Twelve novel BAG-containing visible light-cured resin composites were fabricated using different BAG and monomer configurations (See Table). Four commercially available composites, 2 flowable and 2 conventional, were tested for comparison (see Table). A custom mold was used to make 48 2x2x25mm test bars from each of the composites listed in the Table. Each bar was light polymerized (20 sec/side) using a Bluephase Style LED curing light (Ivoclar Vivadent, Amherst, NY) with irradiance>1000mW/cm2. Samples were aged in RO water for 24 hours at 37°C. Flexural strength (FS) was measured using a 3-point bend test with an ElectroPuls E3000 (Instron, Norwood, MA) (crosshead speed = 0.5mm/min). Data were tested for significant differences using ANOVA and post-hoc Tukey test (α=0.05).
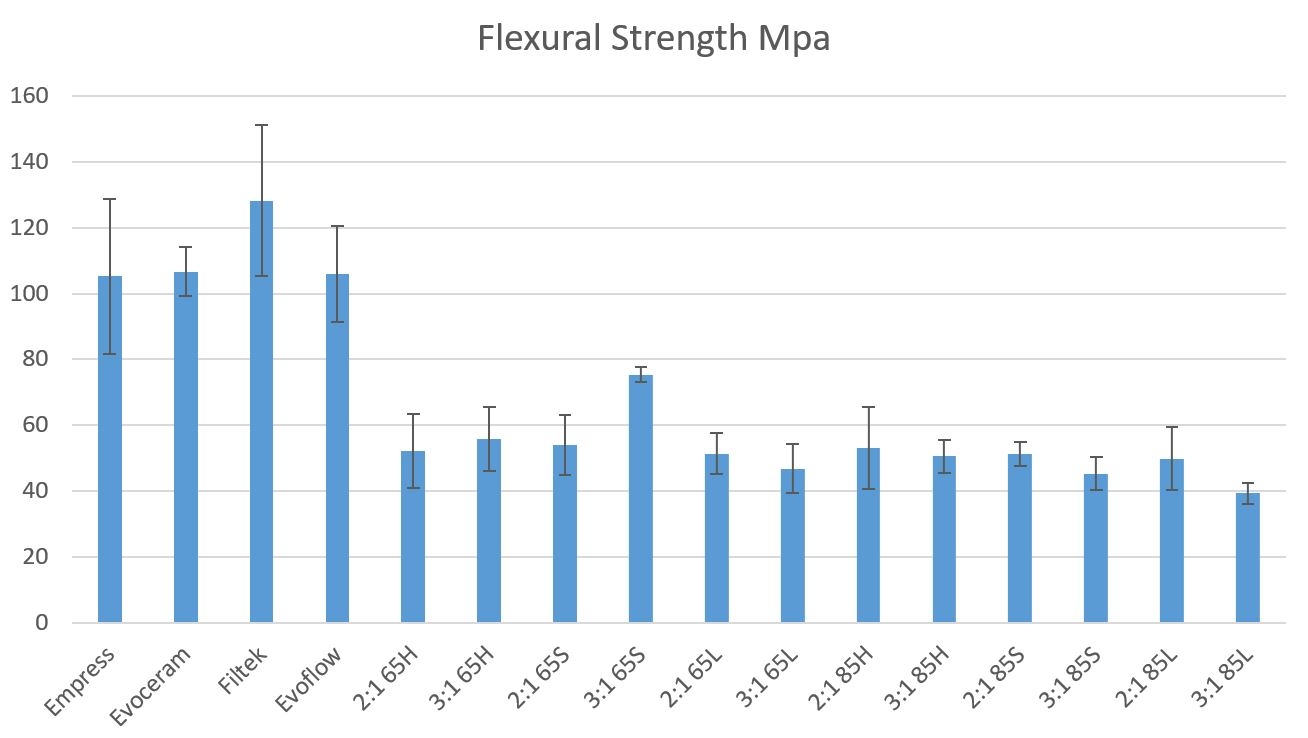
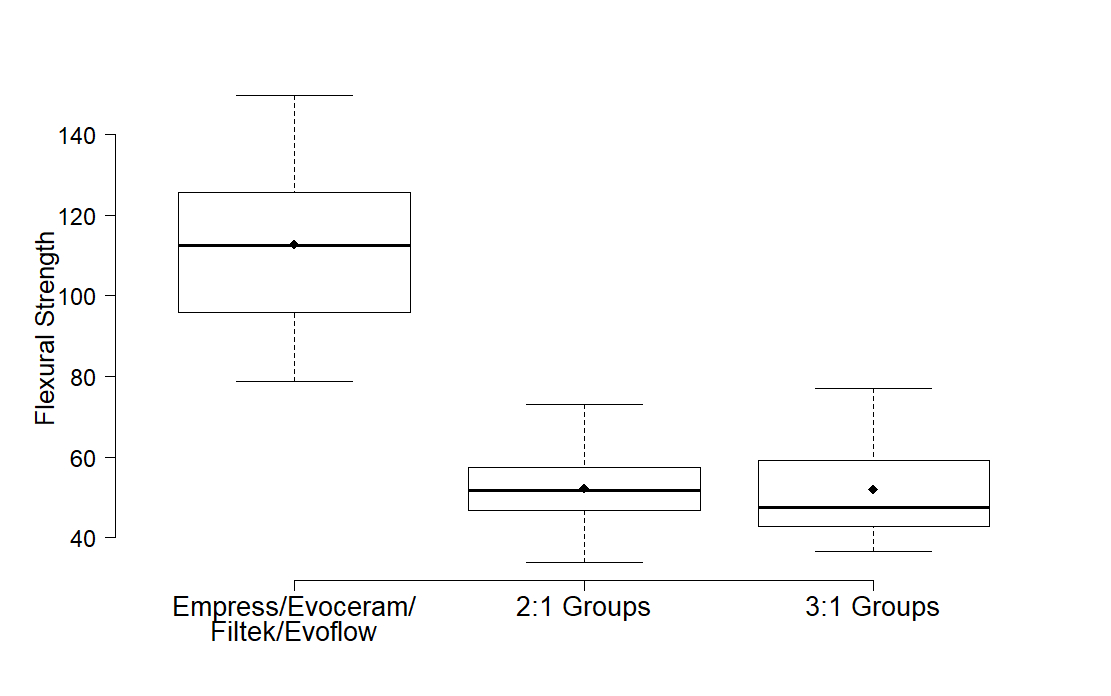
Results: FS results are shown in Figures 1 and 2. Flexural strengths of the commercial composites we tested were statistically similar to each other, but significantly higher (1.4 to 3.25 times higher) than the experimental composites. The strength of the experimental composites did not significantly differ from each other.
Conclusions: Although they had lower flexural strength as tested in this project, composites prepared with antibacterial BAG as a filler may ultimately increase longevity and service life of dental composite restorations. Optimizing the particle size and distribution of the BAG fillers as well as the composition of the monomer mixture will be needed to provide increased strength and handling properties.
Methods: Twelve novel BAG-containing visible light-cured resin composites were fabricated using different BAG and monomer configurations (See Table). Four commercially available composites, 2 flowable and 2 conventional, were tested for comparison (see Table). A custom mold was used to make 48 2x2x25mm test bars from each of the composites listed in the Table. Each bar was light polymerized (20 sec/side) using a Bluephase Style LED curing light (Ivoclar Vivadent, Amherst, NY) with irradiance>1000mW/cm2. Samples were aged in RO water for 24 hours at 37°C. Flexural strength (FS) was measured using a 3-point bend test with an ElectroPuls E3000 (Instron, Norwood, MA) (crosshead speed = 0.5mm/min). Data were tested for significant differences using ANOVA and post-hoc Tukey test (α=0.05).
Results: FS results are shown in Figures 1 and 2. Flexural strengths of the commercial composites we tested were statistically similar to each other, but significantly higher (1.4 to 3.25 times higher) than the experimental composites. The strength of the experimental composites did not significantly differ from each other.
Conclusions: Although they had lower flexural strength as tested in this project, composites prepared with antibacterial BAG as a filler may ultimately increase longevity and service life of dental composite restorations. Optimizing the particle size and distribution of the BAG fillers as well as the composition of the monomer mixture will be needed to provide increased strength and handling properties.