IADR Abstract Archives
Reliability of Manufacturer Stated Irradiance When Selecting Clinical Curing Times
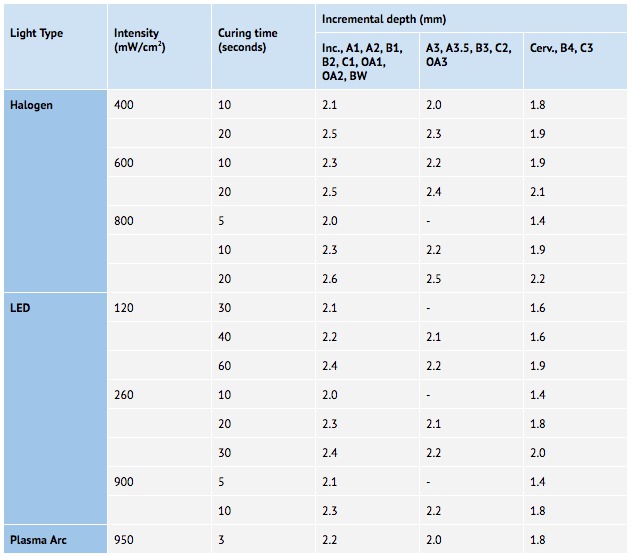
Objectives: This study examined dental Light Curing Units (LCUs) being used in private practice globally to identify whether dentists can rely on the LCU manufacturer stated (MS) irradiance when selecting curing times for their resin-based composite (RBC) materials (See Figure 1).
Methods: LCUs were measured using checkMARC [BlueLight Analytics, Halifax, Canada]; a spectrometer-based device calibrated using a NIST-traceable source. The device recorded light output above 10 hertz over three full curing cycles at 0 mm according to the methods proposed in ISO (DIS) 10650. Also recorded are the LCU model, mode, tip, infection control barrier usage, debris, damage, curing protocols and light cured materials used.
Results: The test results of 40,559 unique LCUs were collected, representing 397 different models of LCUs being used in clinical practice at the time of testing. The 20 (5.0%) most common LCU models, and modes (e.g., standard, high, pulse), made up 26,545 (65.4%) of the tests, and 377 different models represented the remaining 14,014 tests. This study examined 6,437 tests of six commonly used LED LCUs in the most common curing mode (See Table 1. Please note that model names have not been identified so that concerns are not attributed to a specific LCU or manufacturer but focus more on the need for consistent and accurate LCU testing in dental practice).
Conclusions: ● The results show a high degree of variability in the output of LCUs used in clinical practice, both within and between LCU models.
● Without accurate LCU testing, dentists have no way of knowing the output of their LCUs because of a wide variety of factors, including maintenance, damage, and LED degradation.
● Clinical outcomes could be compromised if the MS irradiance is used to determine curing protocols because the RBC intensity and total exposure requirements will not always be satisfied.
Methods: LCUs were measured using checkMARC [BlueLight Analytics, Halifax, Canada]; a spectrometer-based device calibrated using a NIST-traceable source. The device recorded light output above 10 hertz over three full curing cycles at 0 mm according to the methods proposed in ISO (DIS) 10650. Also recorded are the LCU model, mode, tip, infection control barrier usage, debris, damage, curing protocols and light cured materials used.
Results: The test results of 40,559 unique LCUs were collected, representing 397 different models of LCUs being used in clinical practice at the time of testing. The 20 (5.0%) most common LCU models, and modes (e.g., standard, high, pulse), made up 26,545 (65.4%) of the tests, and 377 different models represented the remaining 14,014 tests. This study examined 6,437 tests of six commonly used LED LCUs in the most common curing mode (See Table 1. Please note that model names have not been identified so that concerns are not attributed to a specific LCU or manufacturer but focus more on the need for consistent and accurate LCU testing in dental practice).
Conclusions: ● The results show a high degree of variability in the output of LCUs used in clinical practice, both within and between LCU models.
● Without accurate LCU testing, dentists have no way of knowing the output of their LCUs because of a wide variety of factors, including maintenance, damage, and LED degradation.
● Clinical outcomes could be compromised if the MS irradiance is used to determine curing protocols because the RBC intensity and total exposure requirements will not always be satisfied.