IADR Abstract Archives
Identifying Diagnostic Salivary Biomarkers for Sjögren's Syndrome
Objectives:
Although Sjögren’s syndrome (SjS) is a prevalent autoimmune disorder, confirming the diagnosis remains difficult to achieve due to the lack of definitive diagnostic methods. Development of protein biomarker profile would allow for a highly sensitive and specific diagnostic method. By comparing the proteomes of individuals with and without SjS, alterations in the composition of the salivary proteome can be observed and potential biomarkers identified. The purpose of this study was to determine which proteins could be considered as biomarkers for SjS diagnosis.
Methods:
Twenty-one SjS patients and 18 age-matched healthy controls were recruited. Stimulated whole saliva was collected. One hundred μg of protein was trypsin-digested and alkylated. Digests were desalted for proteomics analysis using nano LC-MS/MS. Multiple search algorithms were used for protein identification. Label-free quantitative analysis was performed using Sieve 2.2 software. For validation, 10 μg of 9 SjS and 9 healthy samples were separated by SDS-PAGE followed by transfer to PVDF membranes for Western blotting. Immunoreactive bands were visualized and quantified using the Odyssey® Infrared Imaging System.
Results:
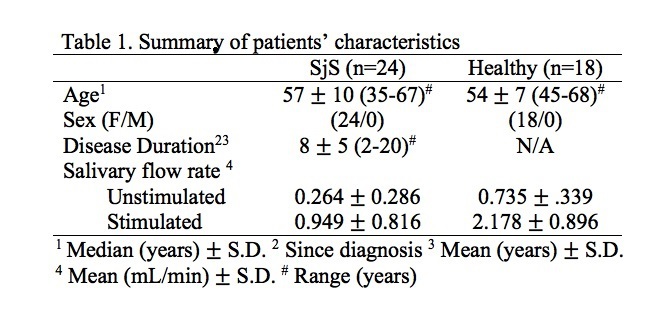
SjS and healthy subjects’ demographics are summarized in Table 1. Unstimulated and stimulated salivary flow rates were lower for patients diagnosed with SjS compared to healthy subjects. Using nano-LC-MS/MS, over 1057 proteins were identified across the samples at a 1% false identification rate. Multiple search engines boosted the identification rate. After chromatographic alignment, MS-features were detected and their peak areas were determined. Thirty-five protein candidates showed significant differences between SjS and healthy samples.
Conclusions:
Candidate proteins were identified that differ in their abundance in stimulated saliva samples collected from SjS and healthy patients. Select candidate proteins are currently being validated by Western blotting to verify whether these proteins can be included in the development of a protein biomarker profile to be used as a non-invasive method of SjS diagnosis.
Although Sjögren’s syndrome (SjS) is a prevalent autoimmune disorder, confirming the diagnosis remains difficult to achieve due to the lack of definitive diagnostic methods. Development of protein biomarker profile would allow for a highly sensitive and specific diagnostic method. By comparing the proteomes of individuals with and without SjS, alterations in the composition of the salivary proteome can be observed and potential biomarkers identified. The purpose of this study was to determine which proteins could be considered as biomarkers for SjS diagnosis.
Methods:
Twenty-one SjS patients and 18 age-matched healthy controls were recruited. Stimulated whole saliva was collected. One hundred μg of protein was trypsin-digested and alkylated. Digests were desalted for proteomics analysis using nano LC-MS/MS. Multiple search algorithms were used for protein identification. Label-free quantitative analysis was performed using Sieve 2.2 software. For validation, 10 μg of 9 SjS and 9 healthy samples were separated by SDS-PAGE followed by transfer to PVDF membranes for Western blotting. Immunoreactive bands were visualized and quantified using the Odyssey® Infrared Imaging System.
Results:
SjS and healthy subjects’ demographics are summarized in Table 1. Unstimulated and stimulated salivary flow rates were lower for patients diagnosed with SjS compared to healthy subjects. Using nano-LC-MS/MS, over 1057 proteins were identified across the samples at a 1% false identification rate. Multiple search engines boosted the identification rate. After chromatographic alignment, MS-features were detected and their peak areas were determined. Thirty-five protein candidates showed significant differences between SjS and healthy samples.
Conclusions:
Candidate proteins were identified that differ in their abundance in stimulated saliva samples collected from SjS and healthy patients. Select candidate proteins are currently being validated by Western blotting to verify whether these proteins can be included in the development of a protein biomarker profile to be used as a non-invasive method of SjS diagnosis.