IADR Abstract Archives
Hypoxia-mimicking Nanofibrous Scaffolds Promote Endogenous Bone Regeneration
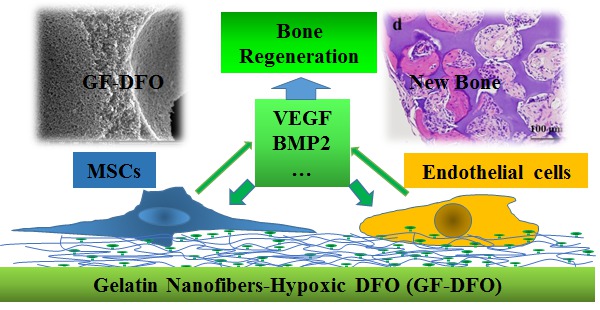
Objectives: Bio-mimetic materials that potentiate endogenous cells or signals to promote bone formation are superior to relying on exogenous cells or signals. Hypoxia promotes bone healing and Deferoxamine (DFO), which is a hypoxia-mimetic agent that chelates Fe3+, encourages healing. However, high cytotoxicity, off-target effects, and short half-life of DFO has significantly impeded its further applications. In the present work, our central hypothesis is that the covalent conjugation of DFO with a polymeric scaffold can decrease its toxicity and elongate its half-life time, and promote bone tissue regeneration through locally activating endogenous hypoxia mediated-angiogenesis and potentially other reparative cells and signals.
Methods: Both 2D and 3D NF-gelatin scaffolds were prepared using thermally induced phase separation (TIPS) method with particle leaching technique (TIPS&P). DFO with different concentrations was crosslinked to NF scaffolds through EDC chemistry. The morphology and microstructure of 2D and 3D GF scaffolds were studied by using Quanta standard Environmental SEM. The cytotoxicity of GF, GF-DFO scaffolds on HUVECs and hMSCs was studied by using MTS assay. The morphologies scaffolds were visualized under the confocal microscopy.The amount of VEGF and BMP2 in supernatant were measured by using human VEGF and human BMP2 ELISA kit. Inbred C57BL/6NHsd male mice were used to create critical-sized cranial bone defect model for in vivo study. Sterile GF and GF-DFO scaffolds (Φ5 mm×1 mm) were directly placed in the cranial defects for 6 weeks and bone formation was analysis by X-ray and histological quantifications.
Results: Our data indicated that DFO-functionalized GF (GF-DFO) scaffolds, with similar micro/macro structures with GF scaffolds, not only demonstrated decreased cytotoxicity on both HUVEC and hMSC, but also significantly increased VEGF expression in vitro. Most importantly, our in vivo experiments in a critical-sized mouse cranial bone defect model showed significant bone growth in most of the GF-DFO scaffolds after 6 weeks, while very little bone growth as observed in the GF scaffolds.
Conclusions: These encouraging data suggested that it is a promising strategy to promote endogenous bone formation by using a hypoxia-mimicking nanofibrous scaffold.
Methods: Both 2D and 3D NF-gelatin scaffolds were prepared using thermally induced phase separation (TIPS) method with particle leaching technique (TIPS&P). DFO with different concentrations was crosslinked to NF scaffolds through EDC chemistry. The morphology and microstructure of 2D and 3D GF scaffolds were studied by using Quanta standard Environmental SEM. The cytotoxicity of GF, GF-DFO scaffolds on HUVECs and hMSCs was studied by using MTS assay. The morphologies scaffolds were visualized under the confocal microscopy.The amount of VEGF and BMP2 in supernatant were measured by using human VEGF and human BMP2 ELISA kit. Inbred C57BL/6NHsd male mice were used to create critical-sized cranial bone defect model for in vivo study. Sterile GF and GF-DFO scaffolds (Φ5 mm×1 mm) were directly placed in the cranial defects for 6 weeks and bone formation was analysis by X-ray and histological quantifications.
Results: Our data indicated that DFO-functionalized GF (GF-DFO) scaffolds, with similar micro/macro structures with GF scaffolds, not only demonstrated decreased cytotoxicity on both HUVEC and hMSC, but also significantly increased VEGF expression in vitro. Most importantly, our in vivo experiments in a critical-sized mouse cranial bone defect model showed significant bone growth in most of the GF-DFO scaffolds after 6 weeks, while very little bone growth as observed in the GF scaffolds.
Conclusions: These encouraging data suggested that it is a promising strategy to promote endogenous bone formation by using a hypoxia-mimicking nanofibrous scaffold.