IADR Abstract Archives
Self-assembling Nanoparticles are Highly Effective Anti-caries Mucosal Vaccines
Objectives: Although anti-caries vaccines have been studied for decades, enhancing the salivary IgA antibody response through mucosal immunization is still a significant challenge. In severe infections caused by S. mutans infection compared with those of the present study, the levels of specific IgA against Antigen I/II (PAc) or glucan-binding regions (GLU) did not simultaneously increase. Thus, the development of anti-caries vaccines is a high priority for preventing dental caries.
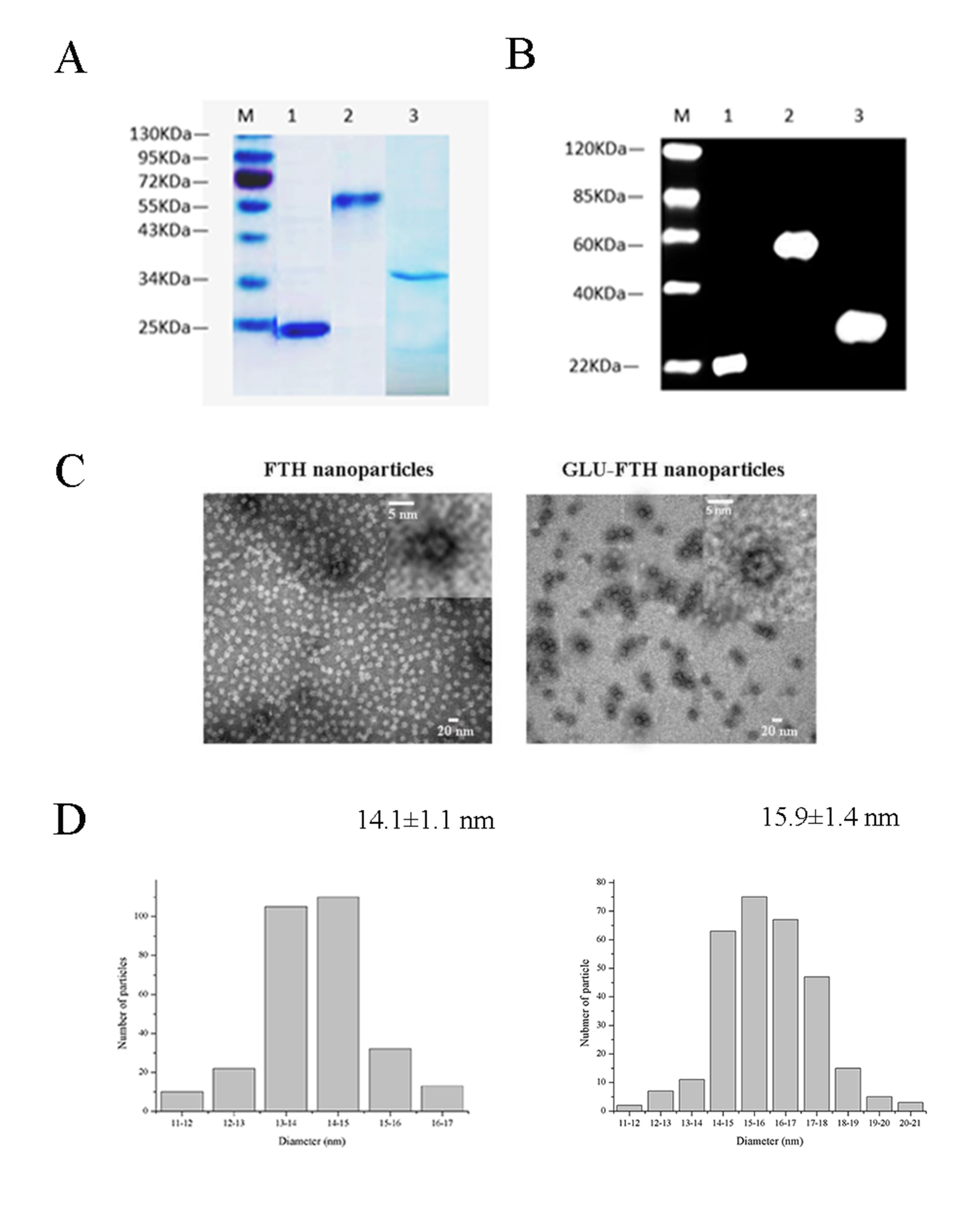
Methods: In the present study, we screened the PAc epitopes and GLU epitopes of streptococcus mutans by bioinformatics which based on the established scientific research platform. We took advantage of the self-assembling function of ferritin by linking the novel epitopic regions from PAc or GLU to the N-terminal exposed area of cage-shaped ferritin. Transmission electron micrographic images were used for the characterization of the novel nanoparticles. We used female BALB/c mice to evaluate the ability of nanoparticles for inducing specific mucosal and systemic responses. Furthermore, the protective efficiency against caries formation in female weanling Wistar rats following intranasal immunization with nanoparticles was compared with the previous anti-caries vaccines we have explored. Further, we evaluated in vitro the phagocytosis of nanoparticles by DCs as well as the ability of nanoparticles to enhance their maturation.
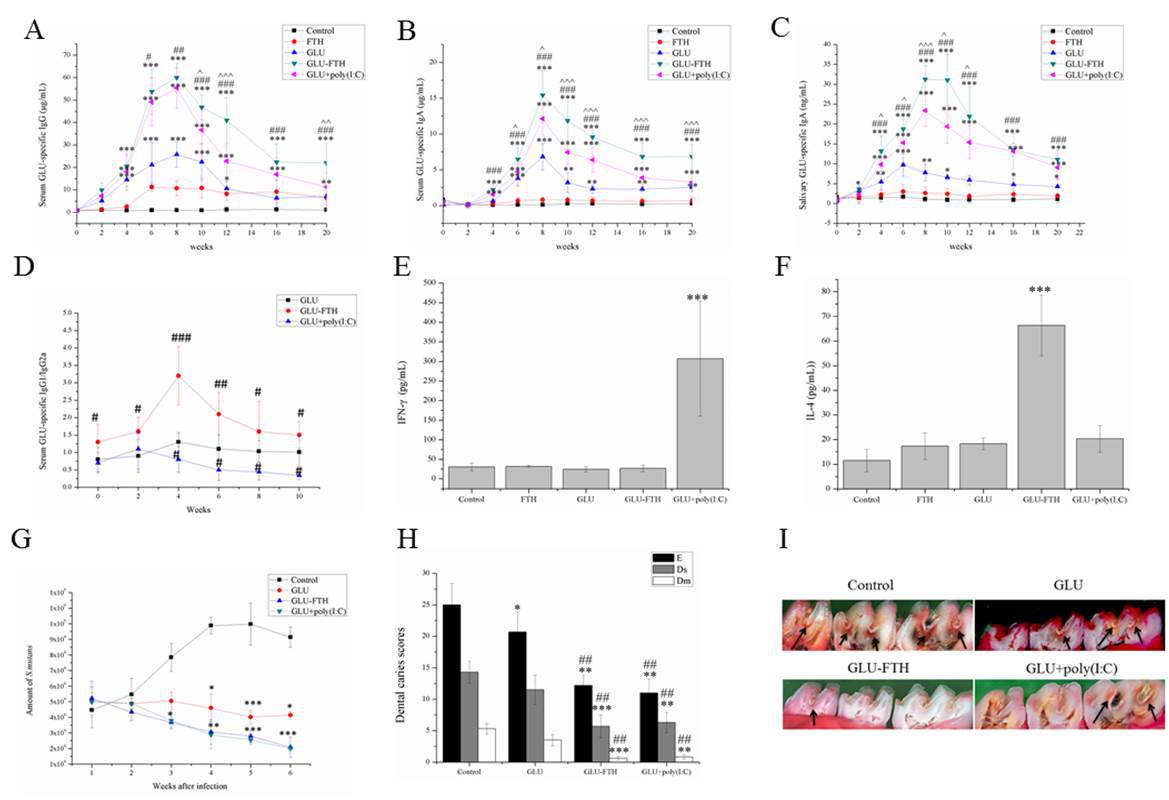
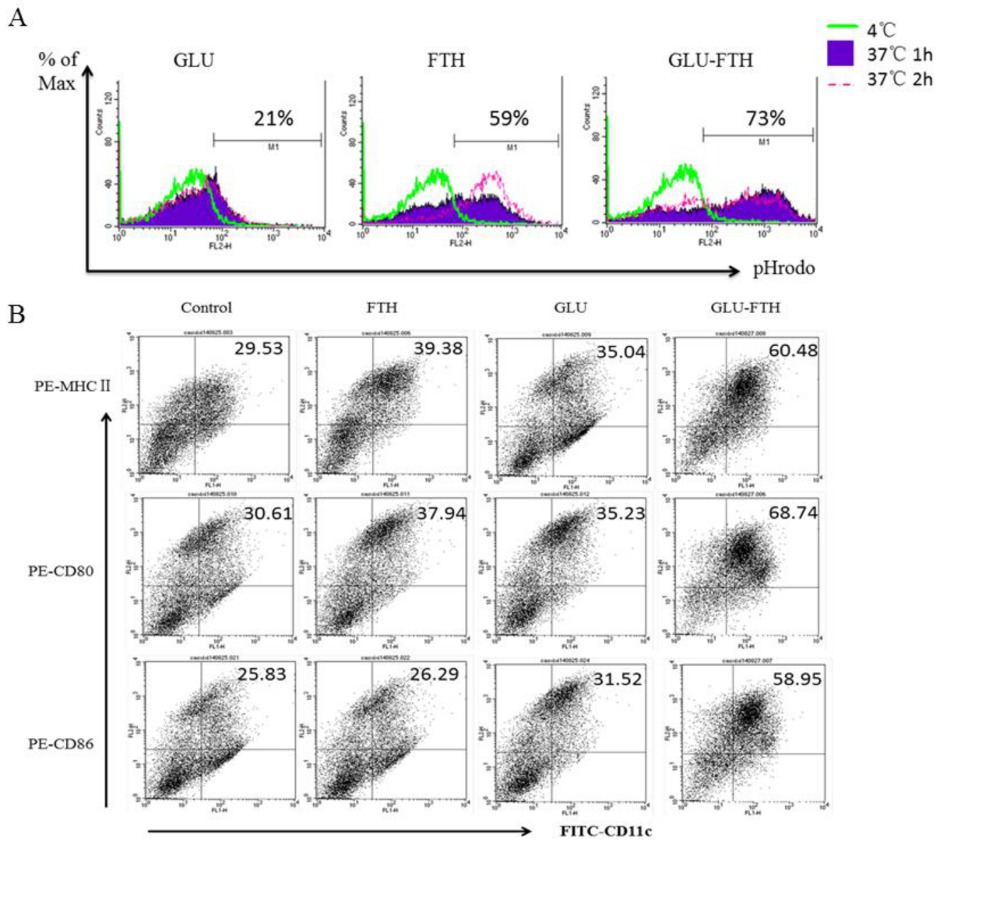
Results: Transmission electron micrographic images of the purified proteins revealed a hollow spherical architecture with a narrow size distribution. Nasal immunization of the novel vaccines induced specific immune reponses and promoted a Th2-biased immune response. In the rat caries model, novel nanoparticles immunization achieved lower caries scores compared with rats immunized with the other vaccine groups. Moreover, the administration of nanoparticles enhanced dendritic cell phagocytosis and maturation.
Conclusions: In summary, the rational design, structural analysis, antigenic profiling, and analyses of the systemic or mucosal immune response presented here establish a novel array of nanoparticles, with potential to serve as components of highly effective anticaries mucosal vaccines.We believe that our findings will inspire a strategy for developing specific mucosal vaccines for other infectious diseases.
Methods: In the present study, we screened the PAc epitopes and GLU epitopes of streptococcus mutans by bioinformatics which based on the established scientific research platform. We took advantage of the self-assembling function of ferritin by linking the novel epitopic regions from PAc or GLU to the N-terminal exposed area of cage-shaped ferritin. Transmission electron micrographic images were used for the characterization of the novel nanoparticles. We used female BALB/c mice to evaluate the ability of nanoparticles for inducing specific mucosal and systemic responses. Furthermore, the protective efficiency against caries formation in female weanling Wistar rats following intranasal immunization with nanoparticles was compared with the previous anti-caries vaccines we have explored. Further, we evaluated in vitro the phagocytosis of nanoparticles by DCs as well as the ability of nanoparticles to enhance their maturation.
Results: Transmission electron micrographic images of the purified proteins revealed a hollow spherical architecture with a narrow size distribution. Nasal immunization of the novel vaccines induced specific immune reponses and promoted a Th2-biased immune response. In the rat caries model, novel nanoparticles immunization achieved lower caries scores compared with rats immunized with the other vaccine groups. Moreover, the administration of nanoparticles enhanced dendritic cell phagocytosis and maturation.
Conclusions: In summary, the rational design, structural analysis, antigenic profiling, and analyses of the systemic or mucosal immune response presented here establish a novel array of nanoparticles, with potential to serve as components of highly effective anticaries mucosal vaccines.We believe that our findings will inspire a strategy for developing specific mucosal vaccines for other infectious diseases.