IADR Abstract Archives
Effect of Mouthrinses on Halitosis: Randomized Crossover Controlled Trial
Objectives: Hydrogen sulfide (H2S) is recently known as a very important gasotransmitter with neuroprotectant, antioxidant, anti-inflammatory, vasodilator, angiogenic and antiapoptotics functions both in physiological and pathological conditions. When methylated, produces methanethiol (CH3SH), and both are the most offensive gases responsible for intra-oral halitosis.
This crossover clinical trial evaluated the effectiveness of chlorhexidine (CHX) or essential oils (EO) based mouthrinses for reduction of H2S and CH3SH levels.
Methods: 21 adult subjects (8 male, 13 female) were randomly allocated into a four period crossover trial. Four mouthrinses were tested: Periogard® with alcohol (positive control) (CHXw/a) and without alcohol (CHXn/a); ListerineTotal® (EOw/a) and ListerineZeroTM (EOn/a) arranged into four sequences of use. All the volunteers were asked to refrain from oral hygiene for a 12-hours period for baseline scores. Each product was used at once, followed by a one-week washout period. The breath was measured by portable gas chromatograph OralChroma™ before rinsing and after 1, 2 and 3 hours. Exploratory analysis of data through measures of central location and dispersion was performed. Comparisons of the means of the variables were made by linear mixed effect model for longitudinal data by using PROC MIXED from SAS® 9.2 software.
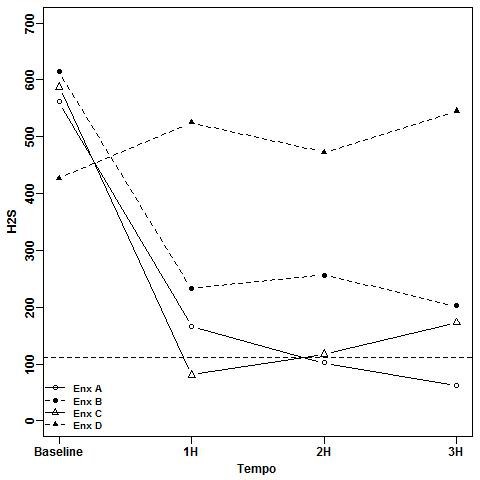
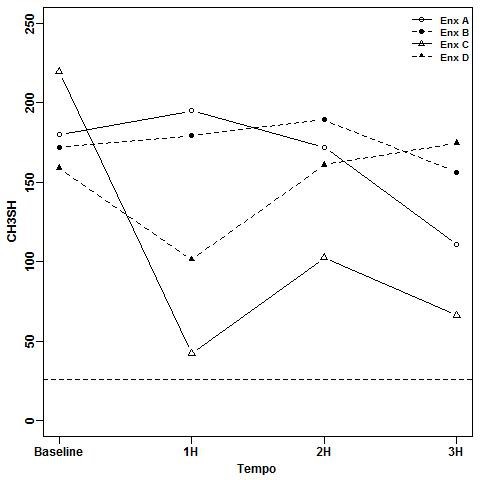
Results: Compared to the baseline, at the first hour, only EOw/a was able to significantly reduce the breath (H2S p<0.0001 and CH3SH p=0.001) for both gases and its effect lasted for up to three hours (H2S p<0.0001 and CH3SH p=0.001). CHXw/a(control) reduced H2S at the first hour (p=0.001) and lasted for three hours (H2S p<0.0001) without effect on CH3SH. CHXn/a just reduced H2S levels but less effectively at the whole period (p=0.001 for the 3 times). EOn/a had no effect on breath, which increased with time for both gases.
Conclusions: The EOw/a presented the best performance against intra-oral halitosis followed by the control CHXw/a and CHXn/a.
This crossover clinical trial evaluated the effectiveness of chlorhexidine (CHX) or essential oils (EO) based mouthrinses for reduction of H2S and CH3SH levels.
Methods: 21 adult subjects (8 male, 13 female) were randomly allocated into a four period crossover trial. Four mouthrinses were tested: Periogard® with alcohol (positive control) (CHXw/a) and without alcohol (CHXn/a); ListerineTotal® (EOw/a) and ListerineZeroTM (EOn/a) arranged into four sequences of use. All the volunteers were asked to refrain from oral hygiene for a 12-hours period for baseline scores. Each product was used at once, followed by a one-week washout period. The breath was measured by portable gas chromatograph OralChroma™ before rinsing and after 1, 2 and 3 hours. Exploratory analysis of data through measures of central location and dispersion was performed. Comparisons of the means of the variables were made by linear mixed effect model for longitudinal data by using PROC MIXED from SAS® 9.2 software.
Results: Compared to the baseline, at the first hour, only EOw/a was able to significantly reduce the breath (H2S p<0.0001 and CH3SH p=0.001) for both gases and its effect lasted for up to three hours (H2S p<0.0001 and CH3SH p=0.001). CHXw/a(control) reduced H2S at the first hour (p=0.001) and lasted for three hours (H2S p<0.0001) without effect on CH3SH. CHXn/a just reduced H2S levels but less effectively at the whole period (p=0.001 for the 3 times). EOn/a had no effect on breath, which increased with time for both gases.
Conclusions: The EOw/a presented the best performance against intra-oral halitosis followed by the control CHXw/a and CHXn/a.