IADR Abstract Archives
Macropinocytosis Uptake Pathways for the Lysyl Oxidase Propeptide (rLOX-PP)
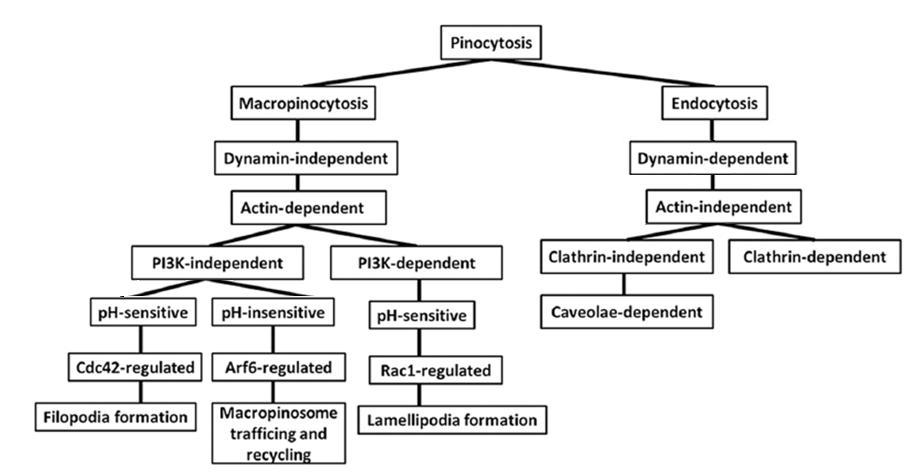
Objectives: LOX-PP is derived from the lysyl oxidase pro-enzyme during the biosynthesis of this important extracellular connective tissue enzyme. LOX-PP has tumor suppressor activity and targets several intracellular proteins. The pathway by which LOX-PP enters cells is unknown and is here investigated and hypothesized to occur by macropinocytosis.
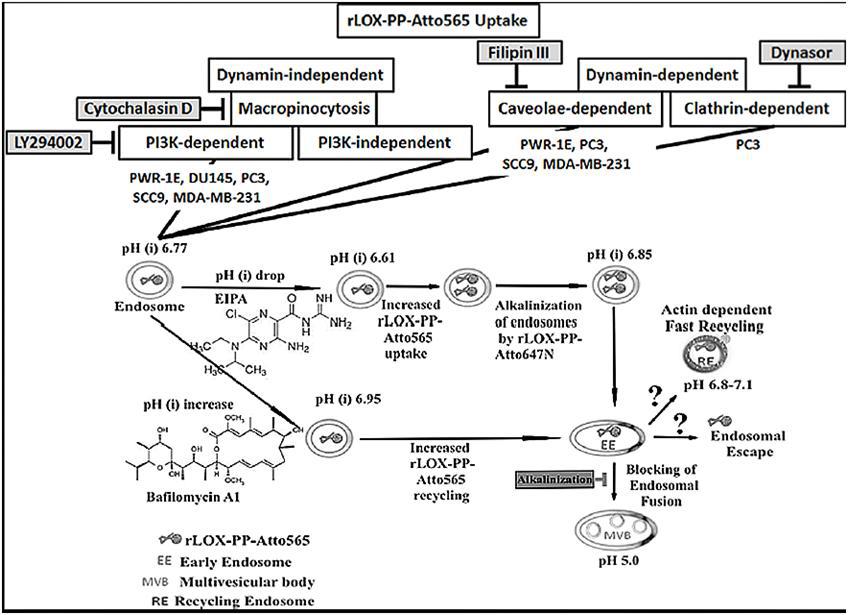
Methods: rLOX-PP was generated, and labeled with ATTO-565 to generate fluorescent rLOX-PP. To determine its cellular localization and quantification and uptake, confocal microscopy and flow cytometry were employed in the presence or absence of several pathway inhibitors in a variety of prostate-, oral- and breast cancer and phenotypically normal cell lines. Endosomal pH change as a function of rLOX-PP-Atto565 was investigated.
Results: Data demonstrated that rLOX-PP enters cells primarily by macropinocytosis in all cell lines tested. rLOX-PP uptake only in DU145 prostate cancer cells appears to occur by a PI3K-independent form of macropinocytosis. Additional LOX-PP uptake pathways occurred to varying degrees in other cell lines tested. Data suggest that caveolae-mediated rLOX-PP uptake occurs in PWR-1E, PC3 and SCC9 cells. PC3 cells also employ clathrin-mediated rLOX-PP uptake. Furthermore, it appears that rLOX-PP buffered both low and high pH endosomes and alkalinized endosomal pH.
Conclusions: That rLOX-PP can be taken up by cells via different pathways may provide opportunities to target cells in specific environments that utilize unique uptake pathways. Both macropinocytosis- and caveola-dependent uptake pathways favor escape from endosomes to the cytoplasm and to known functional LOX-PP targets. Furthermore, the endosomal alkalinizing effect of rLOX-PP may restore Rac1 or PI3K dependent macropinocytosis in cells with a low intracellular pH, increase the uptake and endosomal escape of rLOX-PP in tumors characterized by excess production of lactic acid under hypoxic conditions.
Methods: rLOX-PP was generated, and labeled with ATTO-565 to generate fluorescent rLOX-PP. To determine its cellular localization and quantification and uptake, confocal microscopy and flow cytometry were employed in the presence or absence of several pathway inhibitors in a variety of prostate-, oral- and breast cancer and phenotypically normal cell lines. Endosomal pH change as a function of rLOX-PP-Atto565 was investigated.
Results: Data demonstrated that rLOX-PP enters cells primarily by macropinocytosis in all cell lines tested. rLOX-PP uptake only in DU145 prostate cancer cells appears to occur by a PI3K-independent form of macropinocytosis. Additional LOX-PP uptake pathways occurred to varying degrees in other cell lines tested. Data suggest that caveolae-mediated rLOX-PP uptake occurs in PWR-1E, PC3 and SCC9 cells. PC3 cells also employ clathrin-mediated rLOX-PP uptake. Furthermore, it appears that rLOX-PP buffered both low and high pH endosomes and alkalinized endosomal pH.
Conclusions: That rLOX-PP can be taken up by cells via different pathways may provide opportunities to target cells in specific environments that utilize unique uptake pathways. Both macropinocytosis- and caveola-dependent uptake pathways favor escape from endosomes to the cytoplasm and to known functional LOX-PP targets. Furthermore, the endosomal alkalinizing effect of rLOX-PP may restore Rac1 or PI3K dependent macropinocytosis in cells with a low intracellular pH, increase the uptake and endosomal escape of rLOX-PP in tumors characterized by excess production of lactic acid under hypoxic conditions.